Clinical Pharmacist Leadership in Precision Medicine & PGx
Explore the role of clinical pharmacists in pharmacogenomics (PGx) and targeted drug delivery. Clinical cases, CPIC guidelines, and precision medicine models.
Clinical Pharmacist Role in Precision Medicine
From Pharmacogenomics to Targeted Drug Delivery
Dr. [Name]
Clinical Pharmacist & Precision Medicine Specialist
Genotype → Phenotype → Drug Exposure → Clinical Outcome → Decision → Monitoring
WHY WE ARE HERE
40–60%
of patients do NOT respond to their prescribed drug
15–25%
experience serious adverse drug reactions
WHO | FDA | PharmGKB
The Root Cause
Genetic variation → altered enzyme activity → wrong drug exposure
No PGx testing → empiric dosing → preventable harm
Pharmacist excluded from decision → missed intervention
Genotype → Phenotype → Drug Exposure → Clinical Outcome → Decision → Monitoring
Open with silence. Show stat. Ask: 'How many of your patients are in this 40%?' Pause 10 seconds. Let it land.
Precision Medicine: A Mechanistic Definition
GENOMIC DATA
DNA Sequences & Variants
PHENOTYPE PREDICTION
Enzyme / Target Activity
DRUG SELECTION
Avoidance & Alternatives
DOSE OPTIMIZATION
PK/PD Modifiers
OUTCOME MONITORING
Efficacy & Toxicity Metrics
It's NOT personalized medicine
That's marketing language
It IS variant-driven decision science
Genomic variants predict drug metabolism phenotype
Clinical Pharmacist = The Decision Architect
Translates genomic data into actionable drug therapy decisions
Genotype → Phenotype → Drug Exposure → Clinical Outcome → Decision → Monitoring
Your Clinical Compass
Every case in this session follows this framework
GENOTYPE
Variant detected (e.g., CYP2C19 *2/*2)
PHENOTYPE
Poor Metabolizer predicted
DRUG EXPOSURE
AUC ↑, Clearance ↓
CLINICAL OUTCOME
Toxicity or Therapeutic Failure
DECISION
Pharmacist Intervenes
MONITORING
TDM + Biomarker Tracking
CPIC Level A Evidence | PharmGKB | FDA PGx Biomarker Table
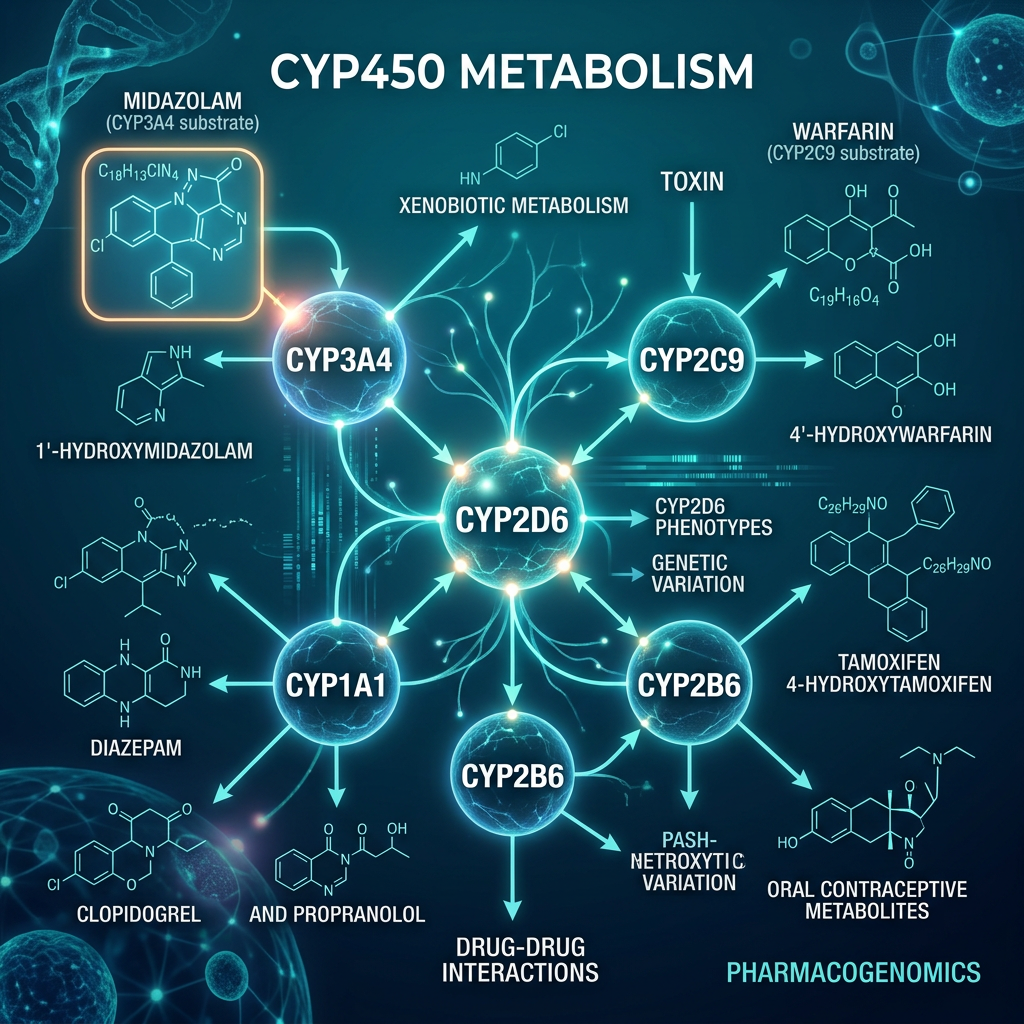
CYP450 Enzyme Variability — The Pharmacogenomic Engine
CYP450: The Drug Metabolism Switchboard
CYP2C19
Clopidogrel, PPIs, Citalopram
13+ allelic variants
CYP2D6
Codeine, Tamoxifen, Antidepressants
100+ allelic variants
CYP2C9
Warfarin, NSAIDs, Phenytoin
IM/PM variants alter bleeding risk
CYP3A4/5
~50% of all drugs
Ethnic variability critical
Pharmacogenomic Mechanism Framework
What Genotype Does to Your PK Parameters
Precision Medicine Symposium
Dr. [Name]
Clinical Pharmacist & Precision Medicine Specialist
Precision Medicine Core Principle: Proactive PK Intervention
CLINICAL CASE 1 — CARDIOLOGY: The Clopidogrel Paradox
CASE 01
52-year-old male. Post-PCI (drug-eluting stent).
On standard Clopidogrel 75mg/day.
Day 30: Stent thrombosis. Readmitted.
Patient was compliant.
Genetic resistance.
GENOTYPE
CYP2C19 *2/*2
Loss-of-function homozygous
Poor Metabolizer
PHENOTYPE
Hepatic CYP2C19 absent
Clopidogrel NOT converted to active thiol metabolite
DRUG EXPOSURE
Active metabolite AUC ↓ 80%
Platelet inhibition: <20% vs target 40-60%
CLINICAL OUTCOME
Stent thrombosis
Mortality risk ↑ 3.5x in *2/*2 carriers
PHARMACIST DECISION
Switch to Ticagrelor 90mg BID
CPIC Level A recommendation
MONITORING
Platelet reactivity units (PRU)<br><span style="font-size: 20px; color: #7DD3FC; margin-top: 8px; display: inline-block;">Target <208 PRU</span>
3-month TEG/PlateletMapping
CPIC Guideline: Clopidogrel/CYP2C19 — Level A | PMID: 22547083
CLINICAL DECISION POINT — YOU DECIDE
CYP2C19 *2/*2 patient. Post-PCI. Day 30 readmission. What is YOUR recommendation?
Continue Clopidogrel + Increase dose to 150mg/day
Physician preference. Patient already on formulary drug.
Switch to Ticagrelor 90mg BID immediately
CPIC Level A. Bypasses CYP2C19. Direct P2Y12 inhibitor.
Add Aspirin 325mg and continue current regimen
Conservative approach. Avoid bleeding risk of switch.
RAISE YOUR HAND — A, B, or C?
30 seconds
Correct Answer: B — CPIC Grade A | Evidence Level: Strong
CLINICAL CASE 2 — ANTICOAGULATION
Warfarin's Genetic Complexity
CASE 02
<strong style="color: white;">67-year-old female.</strong> Atrial fibrillation. Started on Warfarin 5mg/day (standard initiation). Day 7: INR = 6.8. Major epistaxis. Hospitalized. Hematology consult. <span style="color: #FFB300;">Pharmacist reviews: No PGx testing done at initiation.</span>
GENOTYPE
VKORC1 -1639G>A (AA genotype)<br>+ CYP2C9 *2/*3
Double genetic hit
PHENOTYPE
<strong style="color: white;">VKORC1:</strong> ↓↓ Vitamin K epoxide reductase expression (AA = most sensitive)<br><br><strong style="color: white;">CYP2C9 *2/*3:</strong> Impaired warfarin S-enantiomer metabolism
DRUG EXPOSURE
Warfarin clearance ↓ 60-70%
Standard dose = 3x effective dose in this patient
CLINICAL OUTCOME
Supratherapeutic INR 6.8
Major bleeding event. Preventable.
PHARMACIST DECISION
FDA-approved dosing algorithm: VKORC1 + CYP2C9 + CYP4F2 adjusted dose = <strong style="color: #00C896;">1.5mg/day</strong>
EU-PACT, COAG trial evidence
MONITORING
Daily INR first 5 days. Target 2.0-3.0. Genetic-guided stable dose in 2 weeks vs 6 weeks empiric.
The FDA updated Warfarin labeling in 2007 to include PGx. Why is this still not routine in your institution?
CPIC Warfarin Guideline | EU-PACT Trial | PMID: 24251363
CASE 03
CLINICAL CASE 3 — PSYCHIATRY
The Antidepressant Labyrinth
34-year-old female. Major Depressive Disorder. Failed 3 antidepressant trials over 18 months (Sertraline, Fluoxetine, Venlafaxine). Psychiatrist: 'Treatment-resistant depression.'
Pharmacist review: No CYP2D6 testing. Suspicion of pharmacokinetic resistance, not pharmacodynamic.
CYP2D6 *1/*2 xN
Gene duplication × 3. Ultra-Rapid Metabolizer.
Activity Score >2.0
Rapid elimination of all CYP2D6-substrate antidepressants.
Sertraline AUC ↓ 70%
Fluoxetine: active metabolite insufficient. Drug never reaches therapeutic window.
18 Months Misdiagnosed
Treated as treatment-resistant. Unnecessary polypharmacy. Patient stigmatized.
Switch to Mirtazapine
(not CYP2D6 substrate) OR Vortioxetine with dose escalation. CPIC: Avoid CYP2D6-dependent antidepressants in UM.
PHQ-9 Tracking
Monitor depression score at 4, 8, and 12 weeks. Plasma drug level confirmation if needed.
Is it pharmacodynamic or pharmacokinetic resistance? The pharmacist asks the question others don't.
PHARMACIST DECISION FRAMEWORK
CPIC SSRIs/SNRIs Guideline | PharmGKB CYP2D6 Annotations
CASE 04 — PEDIATRIC EMERGENCY
Codeine & The Silent Killer
3-year-old boy. Post-tonsillectomy.
Prescribed Codeine 1mg/kg q4-6h PRN for pain.
Hour 12:
Child found unresponsive. Respiratory rate: 4/min. Pupils: pinpoint. Naloxone administered. ICU admission.
Mother: "He only had 2 doses."
FDA BLACK BOX WARNING (2013)
Codeine contraindicated in pediatric post-tonsillectomy patients.
Ultra-Rapid CYP2D6 metabolizers at lethal risk.
GENOTYPE
PHENOTYPE
DRUG EXPOSURE
CLINICAL OUTCOME
PHARMACIST DECISION
MONITORING
<ul style="margin: 0; padding-left: 20px; color: #cbd5e1; display: flex; flex-direction: column; gap: 14px; font-size: 18px; line-height: 1.4;"><li>CYP2D6 *1/*2 xN</li><li style="color: white; font-weight: 600;"><span style="background: rgba(230,25,43,0.3); color: #ff8a98; padding: 3px 10px; border-radius: 6px; box-shadow: 0 0 10px rgba(230,25,43,0.2);">Ultra-Rapid Metabolizer</span></li><li style="color: #94a3b8; font-size: 16px;">Frequency: 1-2% European, 3-5% African, up to 16% Ethiopian populations</li></ul>
<ul style="margin: 0; padding-left: 20px; color: #cbd5e1; display: flex; flex-direction: column; gap: 14px; font-size: 18px; line-height: 1.4;"><li style="color: white; font-weight: 600;">Codeine → Morphine conversion:<br><span style="color: #ff4d5e; font-size: 21px; font-weight: 700; display: inline-block; margin-top: 6px;">300-400% of normal rate</span></li><li>Massive morphine generation within 2 hours</li></ul>
<ul style="margin: 0; padding-left: 20px; color: #cbd5e1; display: flex; flex-direction: column; gap: 14px; font-size: 18px; line-height: 1.4;"><li>Morphine plasma level:<br><span style="color: #ff4d5e; font-weight: 700; font-size: 21px; display: inline-block; margin-top: 6px;">5-10x expected</span></li><li style="color: white; font-weight: 600;">CNS opioid receptor saturation</li></ul>
<ul style="margin: 0; padding-left: 20px; color: #cbd5e1; display: flex; flex-direction: column; gap: 14px; font-size: 18px; line-height: 1.4;"><li style="color: #ff4d5e; font-weight: 800; font-size: 21px;">Fatal/near-fatal respiratory depression</li><li style="color: #94a3b8;">11 documented deaths reported to FDA (2004-2012)</li></ul>
<ul style="margin: 0; padding-left: 20px; color: #cbd5e1; display: flex; flex-direction: column; gap: 14px; font-size: 18px; line-height: 1.4;"><li style="color: white; font-weight: 600;">Contraindicate codeine in <span style="color: #ff4d5e;">all pediatric post-surgical patients</span></li><li>Substitute: Ibuprofen + Acetaminophen scheduled</li><li><span style="display:inline-block; border: 1px solid #00C896; color: #00C896; background: rgba(0,200,150,0.1); padding: 3px 8px; border-radius: 6px; font-size: 15px; font-weight: 800; margin-bottom: 6px; letter-spacing: 0.5px;">CPIC Level A</span><br>Avoid in UM phenotype</li></ul>
<ul style="margin: 0; padding-left: 20px; color: #cbd5e1; display: flex; flex-direction: column; gap: 14px; font-size: 18px; line-height: 1.4;"><li>If opioid required: Morphine with strict dose titration</li><li style="color: white; font-weight: 600; display: flex; align-items: center; gap: 10px; margin-left: -20px;"><div style="width: 10px; height: 10px; background: #00C896; border-radius: 50%; box-shadow: 0 0 10px #00C896; flex-shrink: 0;"></div> Continuous respiratory monitoring</li></ul>
FDA Drug Safety Communication 2013 | CPIC Codeine Guideline Level A | PMID: 22205192
Precision Medicine Framework
AI vs Pharmacist — Who Wins the PGx Decision?
AI Clinical Decision Support
Patient: CYP2C19 *2/*2 post-PCI
Recommendation: Consider alternative antiplatelet agent.
Suggested: Prasugrel or Ticagrelor.
Confidence: 87%
Source: CPIC Guidelines 2023
Technically correct. Clinically incomplete.
Clinical Pharmacist
✓ Confirms Ticagrelor — but ALSO checks:
Contraindication:
Prior stroke? (TIA history)
Drug interaction:
Concomitant strong CYP3A4 inhibitor?
Patient-specific:
Can afford $180/month vs $8 generic?
Cultural:
Will patient accept daily dual antiplatelet?
Monitoring plan:
PRU testing at 1 month
Educates patient on bleeding signs in Arabic
THE AUDIENCE CHALLENGE
What did the AI miss? Discuss with your neighbor — 60 seconds.
60 SEC
PART II: TARGETED DRUG DELIVERY
PGx Tells Us WHAT to Give.<br>Nano Tells Us HOW to Deliver It.
PGx identifies <strong style="color: white; font-weight: 700;">CYP2C19 *2/*2</strong>: Choose Ticagrelor, not Clopidogrel
Nanodelivery ensures Ticagrelor reaches <strong style="color: white; font-weight: 700;">P2Y12 receptor</strong> with controlled release
<strong style="color: white; font-weight: 700;">Ligand-targeted carriers:</strong> bypass hepatic first-pass for CYP-compromised patients
PGx-Guided Drug Selection + Nano-Optimized Delivery = Precision Pharmacotherapy
Genotype
Drug Selection via PGx
Delivery via Nanotechnology
Optimized Clinical Outcome
Targeted Drug Delivery Systems
Select delivery system based on patient's genotype-predicted metabolism profile.
Nanoemulsions
Oil-in-water droplets (50-200nm). Drug solubilized in lipid core. Enhanced oral bioavailability for poorly soluble drugs.
CYP2C9 PM patients: Cyclosporine nanoemulsion → predictable absorption despite reduced hepatic metabolism.
↑ Bioavailability
↓ Dose variance
Bypasses food effect
Ligand-Targeted
Surface-functionalized nanoparticles. Ligands: monoclonal antibodies, aptamers, folate. Active targeting to overexpressed receptors.
HER2+ breast cancer (ERBB2 variant): Trastuzumab-decorated nanoparticles deliver chemotherapy only to HER2+ cells.
↑ Selectivity
↓ Systemic toxicity
Genotype-matched targeting
Stimuli-Responsive
Release triggered by tumor microenvironment pH (6.5 vs 7.4), enzyme activity, or temperature. Smart release at site of action.
TPMT-deficient patients: pH-triggered 6-MP release in gut avoids systemic toxicity. CPIC guidance for thiopurines.
Site-specific
Enzyme-responsive
Reduces off-target effects
PGx + Nano: The Integrated Precision Pharmacotherapy Model
TOP LEVEL — PGx LAYER
BOTTOM LEVEL — NANO DELIVERY LAYER
Genotype Test
Metabolizer Phenotype
Drug Selection (CPIC)
Dose Calculation
Formulation Choice
Carrier Engineering
Targeting Strategy
Release Profile
OPTIMIZED DRUG EXPOSURE IN TARGET TISSUE
CYP2D6 UM: Increase dose + controlled-release nanocarrier to sustain therapeutic levels
CYP2C9 PM: Reduce dose + standard nanoemulsion for predictable absorption
HER2+ oncology: ERBB2-guided Trastuzumab + ligand-decorated nanoparticle
Clinical Pharmacist = The Integration Architect
⚡ RAPID-FIRE PGx CHALLENGE — ARE YOU READY?
5 HIGH-YIELD MCQs — 10 SECONDS EACH
A CYP2C19 *2/*2 patient on Omeprazole. What is expected?
A) Reduced acid suppression
B) Enhanced acid suppression
C) No change
D) Increased gastric bleeding
Q2: CYP2D6 UM + Codeine = ?
Q3: VKORC1 AA + Standard Warfarin = ?
Q4: Best delivery for CYP2C9 PM patient = ?
Q5: CPIC Level A for CYP2D6 + Codeine recommends = ?
10
Use your clinical reasoning. No notes. No phones. Just you and your training.
Traditional
Pharmacist Role
Dispensing
Drug Interaction Checker
Dose Calculator
Supporting Role
Reactive
Waits for orders
Clinical Pharmacist
in Precision Medicine
PGx Test Interpreter
Genotype-Guided Drug Selector
Therapeutic Drug Monitor
Nano-Delivery Consultant
Decision Authority
Proactive Intervener
Prevents harm before it happens
The clinical pharmacist who integrates PGx + nanotechnology is not a support role. They are the precision medicine architect.
The PGx Clinical Workflow
From Test to Monitoring
TEST
Genetic Testing
INTERPRET
Pharmacist Interpretation
DECIDE
Clinical Decision
MONITOR
Therapeutic Monitoring
Clinical Pharmacist leads Steps 2, 3, and 4.
REGIONAL LEADERSHIP
The MENA Advantage
Infrastructure gaps are opportunities for nations that move first
Infrastructure Gap = Opportunity
No legacy EHR to replace. Libya & MENA can build PGx-integrated systems from scratch. First-mover advantage in national PGx registry.
Genetic Uniqueness = Scientific Value
North African populations underrepresented in global PGx databases. CYP2D6 UM prevalence up to 3-5× higher than European. Unique founder alleles with global research value.
Academic Institutions as Launch Pads
Universities of Tripoli, Benghazi as PGx implementation centers. Train next-gen clinical pharmacists. Hospital-university partnerships.
FEASIBLE NOW — Pilot Model
Select cardiac/anticoagulation clinic
Partner with 1 lab for CYP2C19/VKORC1 genotyping
Assign clinical pharmacist as PGx interpreter
50 patients/month — track INR events & ADRs
Startup cost: ~$5,000 | ROI: 2 prevented hospitalizations = break even
Precision Medicine Symposium
Implementation Roadmap — Building a PGx-Ready Institution
Precision Medicine Implementation: From Zero to Clinical Practice
FOUNDATION
Month 1-3
Identify champion pharmacist + physician
Select 2-3 PGx genes (CYP2C19, VKORC1, CYP2D6)
Partner with genotyping laboratory
Select patient population (cardiology/anticoag)
$$ Low
PILOT
Month 4-9
Enroll 50-100 patients
Implement CPIC-based decision protocols
Track: ADR rates, hospitalizations, TTR for warfarin
Build PGx database — first MENA cohort
$$$ Moderate
SCALE
Month 10-18
Expand to oncology, psychiatry
Train 5+ pharmacists in PGx interpretation
Integrate into hospital EHR
Publish outcomes — MENA-first data
$$$$ Strategic investment
LEAD
Year 2+
National PGx registry
Regional MENA collaboration network
Clinical pharmacy PGx certification program
Pharmaceutical industry partnerships
Investment → Revenue
Barrier: Cost
Solution: Start with high-impact, low-cost genes (CYP2C19)
Barrier: Expertise
Solution: Online CPIC certification. PharmGKB free tools.
Barrier: Physician Buy-in
Solution: Show one prevented ADR. Data convinces.
Precision Medicine Framework
The Evidence Foundation
Every recommendation in this presentation is evidence-graded
Clinical Guidelines
Key Clinical Trials
Nanomedicine Resources
CPIC — Clinical Pharmacogenomics Implementation Consortium | cpicpgx.org | Level A-D evidence grading
PharmGKB — Pharmacogenomics Knowledgebase | pharmgkb.org | Gene-drug annotations
FDA PGx Biomarker Table | 300+ drug-gene pairs | Updated 2024
Dutch Pharmacogenetics Working Group (DPWG) | Complementary to CPIC
EU-PACT Trial (NEJM 2013) — Genotype-guided warfarin dosing | PMID: 24251363
TRITON-TIMI 38 — CYP2C19 & Clopidogrel outcomes | PMID: 19106084
GeneSight Trial — PGx in psychiatry reduces ADRs 30% | PMID: 31960068
COAG Trial — Warfarin PGx dosing | PMID: 24251363
Codeine Safety FDA Communication 2013 | PMID: 22205192
Torchilin VP. Nat Rev Drug Discov. 2014 — Nanocarrier drug delivery
Petros RA, DeSimone JM. Nat Rev Drug Discov. 2010 — Nanoparticle design
Peer D et al. Nature Nanotechnology 2007 — Cancer targeting
Duncan R. Nat Rev Cancer 2006 — Polymer therapeutics
AlSiraj Y et al. MENA PGx studies 2021 — Arab population CYP variants
All CPIC guidelines are freely accessible at cpicpgx.org | PharmGKB annotations at pharmgkb.org | FDA table: fda.gov/drugs/science-research-drugs/table-pharmacogenomic-biomarkers-drug-labeling
Leave This Room. Change One Thing Tomorrow.
5 Concrete Actions. No Exceptions.
01
Before prescribing a CYP2C19/2D6/2C9 substrate — ASK: Has this patient been genotyped?
START TODAY
02
Review your anticoagulation patients. Identify those on empiric warfarin without PGx testing.
THIS WEEK
03
Complete 1 free CPIC module at cpicpgx.org — Certify yourself in CYP2C19 or VKORC1
THIS MONTH
04
Find 1 physician ally. Show them one PGx case. Build the bridge.
THIS MONTH
05
Propose a 50-patient PGx pilot. Write the 1-page protocol tonight.
THIS QUARTER
"Precision medicine does not begin with a government program. It begins with a single pharmacist who decides: Not on my watch."
Precision Medicine Symposium
"The genome doesn't lie. The drug doesn't care about your protocol. Only the clinical pharmacist — armed with both — can bridge the gap between what the drug is supposed to do, and what it actually does in this patient, at this dose, right now."
— Precision Medicine Principle
Genotype → Phenotype → Drug Exposure → Clinical Outcome → Decision → Monitoring
[Email]
[LinkedIn]
Clinical Pharmacist | Precision Medicine | PGx | Targeted Delivery
- pharmacogenomics
- precision-medicine
- clinical-pharmacist
- pgx
- targeted-drug-delivery
- nanotechnology
- cpic-guidelines
- pharmacokinetics