Besifloxacin-Eluting Contact Lens Review: Sustained Delivery
A comprehensive review of besifloxacin-eluting contact lenses for treating bacterial keratitis, featuring 21.6x higher drug bioavailability than eye drops.
JOURNAL CLUB PRESENTATION
Besifloxacin-Eluting Contact Lens
with Sustained Drug Delivery and Enhanced Bioavailability
Kuang L, Boychev N, Chen L, Ross AE, Kanu LN, et al.
International Journal of Pharmaceutics: X, 11 (2026) 100511
Presenter 1
Presenter 2
Presenter 3
Presenter 4
Presenter 5
Harvard Medical School / Mass Eye and Ear
Presentation Overview
Introduction & Background
Problem Statement
Besifloxacin: The Drug
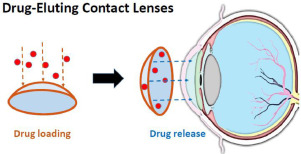
Drug-Eluting Contact Lens Concept
Materials & Methods — Fabrication
Physicochemical Characterization
In Vitro Drug Release
Stability Studies
Antimicrobial Efficacy & Cytotoxicity
Pharmacokinetics & Biocompatibility
Comprehensive Journal Club Review | Masters Level Evaluation
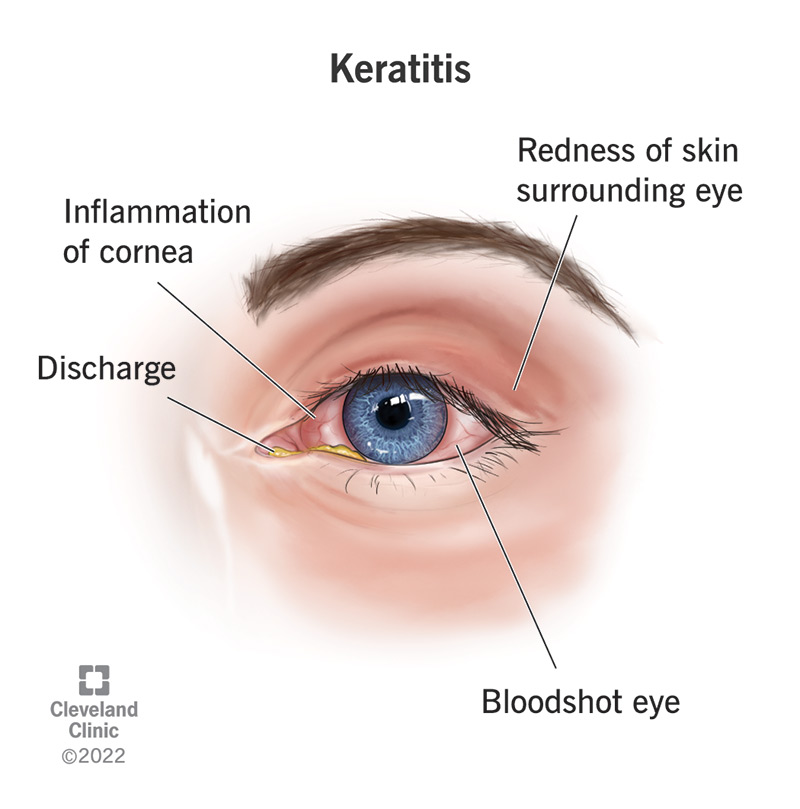
Introduction: Bacterial Keratitis
What is Bacterial Keratitis?
A rapidly progressive ocular infection caused by bacterial pathogens. Leading cause of corneal blindness worldwide.
Prevalence & Impact
Significant global burden; affects vision and quality of life. Requires urgent and effective treatment.
Current Standard of Care
Intensive topical antibiotics (eye drops or ointments) administered hourly, even at night.
Key Clinical Limitation
Ocular bioavailability typically <7%. Poor corneal penetration + patient non-compliance.
McDonald et al., 2014 | Ung et al., 2019
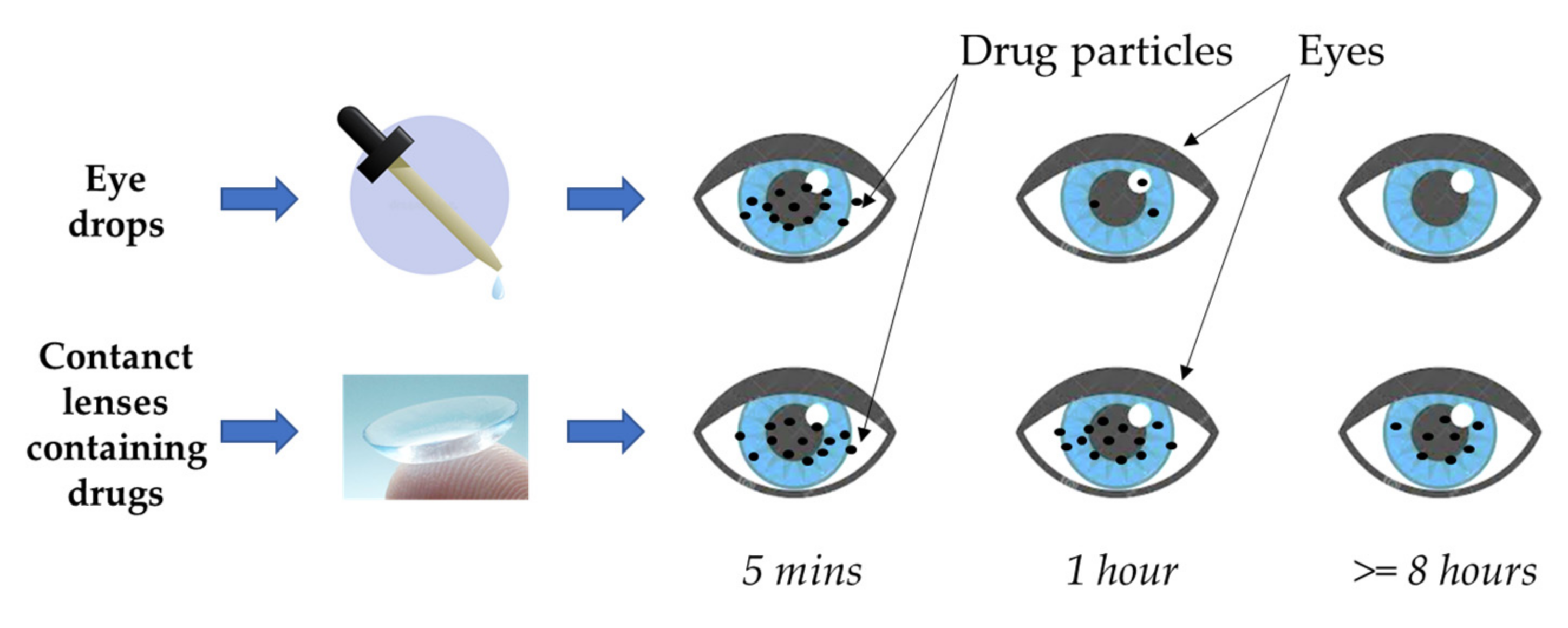
Barriers to Ocular Drug Delivery
Corneal Layers
Hydrophobic epithelium + endothelium flank the hydrophilic stroma — impedes both hydrophobic & hydrophilic molecules
Tear Turnover
0.5–2.2 μL/min tear turnover dramatically reduces drug concentration
Nasolacrimal Drainage
Drug washed away via nasolacrimal duct
Blinking & Reflex Lacrimation
Further reduces residence time
Drug Half-Life in Tear Film
Only 2–5 minutes for most conventional eye drops!
Result: Intensive hourly dosing regimens required → poor compliance → compromised efficacy
Ross et al., 2019 | Peng et al., 2022 | Jumelle et al., 2020
Besifloxacin (BXF): Drug Profile
Drug Class
Pharmacochemical Properties
Why is BXF Challenging to Deliver?
Tótoli & Salgado, 2018 | Comstock et al., 2010
Existing Formulation Strategies & Their Limitations
Strategy
Examples
Improvement
Limitation
Viscosity enhancers / mucoadhesives
DuraSite® (Besivance®)
Extended ocular residence
Impaired corneal penetration, blurred vision, stickiness
Lipid-based nanocarriers
Liposomes, NLC, NLCs
Improved drug loading & penetration
Sustaining therapeutic levels remains challenging
In situ forming gels
Chitosan-gelrite systems
Better retention
Short-term release, discomfort
Polymeric microneedles
PLGA microneedle arrays
Direct corneal drug delivery
Requires professional administration
Nanofibrous ocular inserts
BXF-loaded nanofibers
Extended release
Sub-therapeutic concentrations
Drug-eluting Contact Lenses
Present Study (BXF-CL)
Sustained 24h+ release, 21.6x higher Cmax
— (This study addresses prior gaps!)
Gap: No besifloxacin-eluting contact lens system had been reported prior to this study
dos Santos et al., 2020 | Polat et al., 2022 | Bhatnagar et al., 2018 | Peng et al., 2022
Study Objectives & Hypothesis
A besifloxacin-eluting contact lens (BXF-CL) incorporating an EC/RSPO polymer film reservoir can overcome the physicochemical limitations of besifloxacin to provide sustained therapeutic ocular drug delivery exceeding conventional eye drops.
Fabricate BXF-CL
Develop and optimize drug-loaded polymer film embedded within contact lens periphery
Physicochemical Characterization
Assess morphology, water content, light transmittance
In Vitro Drug Release
Evaluate sustained-release kinetics vs commercial contact lenses
Stability Assessment
Evaluate drug integrity after UV curing, sterilization (autoclave & gamma irradiation), and 2-year storage
Antimicrobial Efficacy
Test against S. aureus and E. coli using agar disk diffusion
In Vivo Pharmacokinetics
Compare aqueous humor drug levels vs hourly Besivance® eye drops in rabbits
First-ever besifloxacin-eluting contact lens system reported in literature
Materials & Methods: Overview
Key Materials
Methods Overview
All in vivo protocols approved by IACUC — Schepens Eye Research Institute
BXF-CL Fabrication Process
Drug-Polymer Solution
BXF + EC + RSPO dissolved in HFIP
Low-dose: 1:5 (BXF:Polymer)
High-dose: 2:5 (BXF:Polymer)
EC/RSPO blend ratio = 1:1
Spin Coating
50 μL pipetted onto concave blank
Spun at 100 rpm for 6 min
Vacuum dried in desiccator x 7 days
Central Aperture
4-mm dermal biopsy punch used
Creates crisp central aperture
Maintains optimal optical clarity
UV Polymerization
350 μL liquid methafilcon added
Fully covers the dried polymer film
UV cured at ~110 mW/cm² for 5 min
Lathing
Solid methafilcon cylinder is lathed
Machined into final contact lens shape
Final BXF-CL
Hydrated lens OD: 14.7 mm
Polymer film OD: 12.0 mm
Central aperture: 5.2 mm
Film thickness: 51–60 μm
Polymer Matrix Rationale
hydrophobic, diffusion-limiting matrix → controls release rate
flexible, tunable permeability → prevents cracking
optimal film integrity + encapsulation performance
Why peripheral film placement?
Preserves clear central optical zone during drug delivery.
Mimics structural aperture design of cosmetic tinted contact lenses.
Physicochemical Characterization
Morphology (OCT)
Light Transmittance
Water Content
✅
BXF-CLs are physically and optically comparable to commercial contact lenses
Source: Section 3.1 | Chen et al., 2022 | Nguyen et al., 2021
In Vitro Drug Release Kinetics
* p = 0.015–0.029 (Mann-Whitney U) vs Commercial CL | Source: Section 3.2
Commercial CL: Burst Release
29.7 ± 1.3 μg (83%) within 1st hour
No drug detected after 4h
Total: only 35.8 ± 2.3 μg/lens
BXF-CLs: Sustained Release
Sustained and steady release over 24h
Low-dose: 276.7 μg (96.6% of drug load)
High-dose: 521.7 μg (97.7% of drug load)
Key Achievement
Up to 15.4× greater total drug delivery vs commercial CLs
Assay conditions: PBS (pH 7.4), 37°C, 5 mL sink conditions, HPLC quantification
Stability Studies:
UV Irradiation & Sterilization
UV Irradiation Stability
Drug-polymer film exposed to same UV conditions as encapsulation (~110 mW/cm², 5 min)
Interpretation: Polymer matrix PROTECTS besifloxacin from UV damage during fabrication
BXF within polymer film
Identical UV-Vis peaks (245, 288, 338 nm) + identical HPLC retention time (10.1 min)
→ NO DEGRADATION
Free BXF solution (control)
Loss of characteristic peaks
→ Significant degradation
Autoclaving (121°C, 220 kPa, 20 min)
Drug itself: stable (no degradation) ✅
BUT: 58% (low) & 49% (high) cumulative release DECREASED ❌
Drug leached into autoclave buffer during sterilization
Release duration shortened from 48h to 12h
~41% drug released within 1st hour
Conclusion: AUTOCLAVING NOT SUITABLE for BXF-CLs ❌
Gamma Irradiation (25 kGy, Cobalt-60)
Cumulative release: PRESERVED (p = 0.40 low, p = 0.90 high) ✅
Similarity factor f₂ = 91.7 (low) & 81.8 (high) — both ≥50 = SIMILAR ✅
LC-MS/MS: drug integrity confirmed ✅
Morphology, transmittance, water content: all unchanged ✅
✅ GAMMA IRRADIATION SELECTED as terminal sterilization method
Source: Section 3.3 | Galante et al., 2018 | Shah et al., 1997
Long-Term Stability: 2-Year Storage Study
Study Design
Storage conditions: Dry state, 4°C, amber glass vials, preservative-free
Duration: 2 years
n = 5 lenses per formulation per time point
Evaluated: Drug release kinetics, morphology, light transmittance, water content
Drug Chemical Integrity
HPLC: drug released from stored lenses identical to pristine besifloxacin. No degradation detected.
Cumulative Release Amount
Low-dose: p = 0.27 → NOT significantly different from baseline (f₂ = 80.3)
High-dose: p = 0.54 → NOT significantly different from baseline (f₂ = 83.0)
Both f₂ values ≥50 → Release profiles are SIMILAR ✅
Lens Morphology & Transmittance
All lenses maintained normal morphology
Light transmittance: p = 0.27 (low) / p = 0.48 (high) → unchanged
Water content: p = 0.45 (low) / p = 0.78 (high) → unchanged
Clinical & Commercial Implication
Extended 2-year shelf life enables commercial viability and clinical practicality
Dry storage at 4°C is feasible for medical-grade preservation
BXF-CLs maintain drug integrity, release kinetics, and lens properties for at least 2 years — supporting strong translational potential
Source: Section 3.3.3 | Kamaly et al., 2016
In Vitro Antimicrobial Efficacy
Methods & Principle
Agar diffusion & HPLC concentrations are CONSISTENT (p=0.17–0.43, Kruskal-Wallis) — confirming bioactive drug integrity
High-dose > Low-dose
Drug Bioactivity Confirmed
Besifloxacin retained full antimicrobial potency after fabrication, sterilization, and release
Gram+ & Gram− Coverage
Effective broad-spectrum antibacterial activity preserved in BXF-CL system
Clinical Significance
BXF-CLs release therapeutically active drug concentrations throughout 8h wear — sufficient for keratitis treatment
Section 3.4 | Comstock et al., 2010 | Kuang et al., 2021
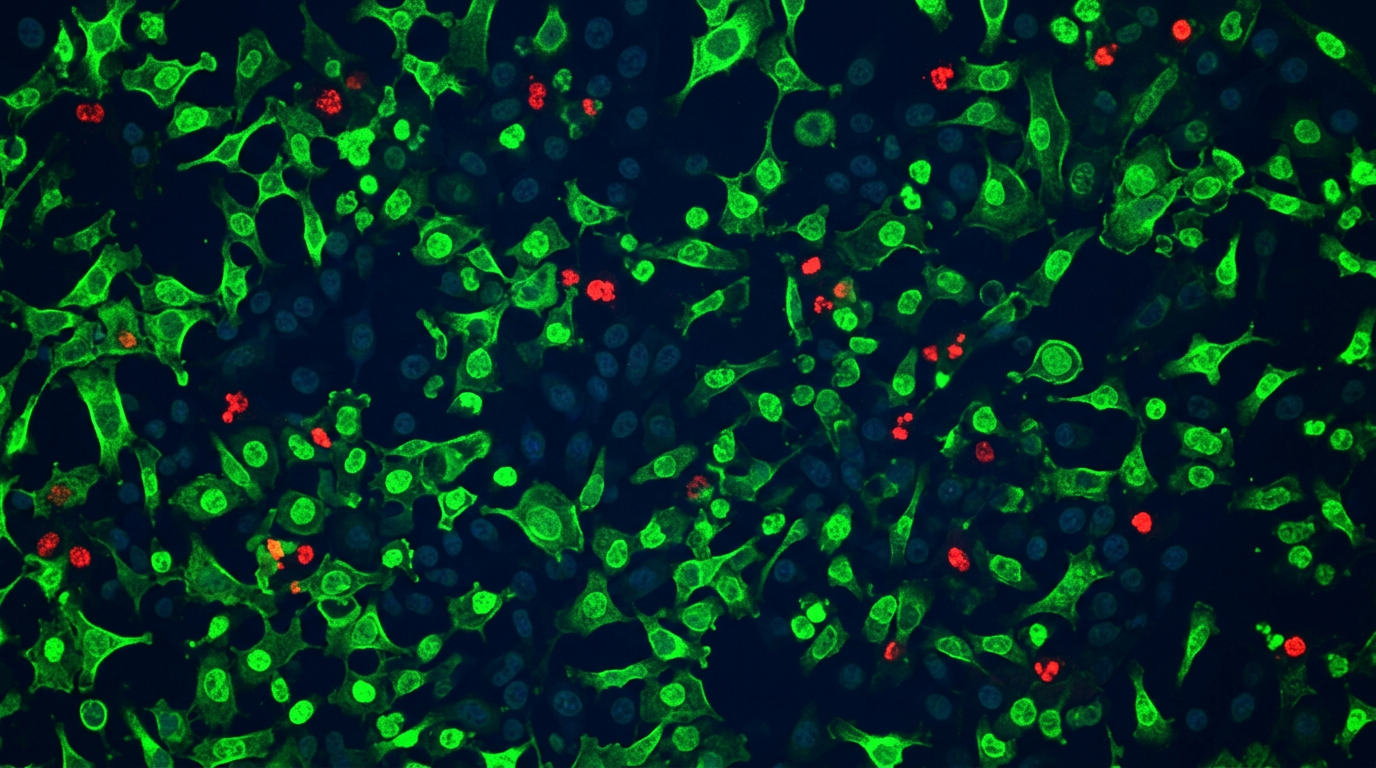
In Vitro Cytotoxicity on Human Corneal Epithelial Cells
Section 3.5 | ISO 10993-5:2009 | Baig et al., 2020
Ocular Pharmacokinetics: Study Design
Section 2.13 | Ross et al., 2019 | Kuang et al., 2021
6 NZW Rabbits (non-terminal crossover)
Permanent lateral tarsorrhaphy
7-day healing period
ARM A: BXF-CL GROUP
Fresh lens applied at each timepoint
ARM B: EYE DROP
Besivance® 0.6% 5 drops over 4h
Aqueous Humor Collection 1h, 2h, 4h, 8h, 12h, 24h (30G needle, 100 μL)
3-day washout between arms
HPLC drug quantification
Rationale for Aqueous Humor Sampling
Located immediately posterior to cornea
Topical drugs enter predominately via cornea (~90% penetration — Doane et al., 1978)
Provides a conservative estimate of actual corneal drug levels
Allows non-terminal crossover design → reduces animal use (3Rs principles)
Rabbits: Ocular dimensions closely approximate human eyes
Eye Drop Dosing Protocol
5 drops administered hourly (doses at 0, 1, 2, 3, 4h)
Dosing stopped after 5th drop — concentrations plateau after 3–4 doses
Simulates a robust 24-drop daily regimen (AUC estimated)
Note: Each drop = one standard clinical dose of Besivance® 0.6%
Pharmacokinetic Results: BXF-CLs vs Hourly Eye Drops
Source: Table 2 | Section 3.6 | Kuang et al., 2021 | Mah & Sanfilippo, 2016
Bioavailability Enhancement & Drug Utilization Efficiency
AUC Comparison — Bioavailability Marker
Drug Dose Efficiency
Why Better Bioavailability?
Clinical & Stewardship Implications
Source: Section 4 | Ciolino et al., 2014 | Lanier et al., 2020
In Vivo Biocompatibility & Ocular Safety
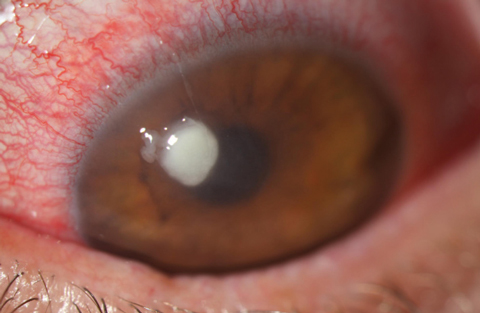
Draize Test (Ocular Irritation Study)
Study Design
Draize Test Results
BXF-CLs show NO ocular irritation potential — non-irritative material classification
In Vivo Lens Wear Safety (48h Clinical Monitoring)
Assessment Parameters
Fluorescein Staining
BXF-CLs confirmed safe and biocompatible in both chemical safety (Draize) and in vivo wear assessments
Source: Section 3.7 | Wilhelmus, 2001 | Bengani et al., 2020
Discussion
: Mechanism & Clinical Significance
Why BXF-CL Works: Mechanistic Insights
Overcoming BXF's Dual Challenge
Post-Lens Tear Film Effect
Dose Efficiency
Clinical Advantages vs Alternatives
BXF-CL combines the convenience of self-administration with sustained, preservative-free release — a clinically adaptable platform
Source: Section 4 | Stone et al., 2009 | Polat et al., 2022
Limitations & Future Directions
Dry Storage Requirement
Current BXF-CL must be stored dehydrated → requires pre-application hydration step
Implication: Best suited for supervised clinical/in-office use, not home use
Solution needed: Evaluate stability in hydrated packaging for future iterations
E. coli as Gram− Model (Not P. aeruginosa)
P. aeruginosa is the most prevalent Gram− in bacterial keratitis
E. coli was chosen for: wider ZOI dynamic range + better quantitative precision
P. aeruginosa has higher intrinsic resistance + restricted agar diffusion → less precise calibration
Limitation: Efficacy against P. aeruginosa not yet demonstrated
Animal Model Only — No Human Clinical Data
Pharmacokinetics validated only in rabbit model (ocular dimensions similar to humans)
Phase I/II/III clinical trials needed for regulatory approval
Translation studies required
In Vitro Efficacy Only — No In Vivo Infection Model
No bacterial keratitis animal model used in this study
Proof-of-concept antimicrobial efficacy shown; in vivo efficacy studies needed
Hydrated Packaging Studies
Evaluate BXF-CL stability and drug retention during long-term storage in hydrated state
Enables consumer convenience and at-home use
P. aeruginosa Efficacy Testing
In vitro and in vivo testing against the primary Gram− pathogen in bacterial keratitis
In Vivo Infection Efficacy
Rabbit model of bacterial keratitis (MRSA-keratitis model as established precedent)
Validate therapeutic efficacy against eye infection in animal models
Clinical Translation
Phase I safety trials → Phase II efficacy studies
Regulatory pathway (FDA, CE Mark)
Manufacturing scale-up and commercialization studies
These limitations are acknowledged transparently
future studies are clearly defined and clinically motivated
Source: Section 4 Discussion | Sanders et al., 2009
Conclusion & Key Takeaways
SUMMARY
BXF-CLs represent a clinically adaptable platform for sustained ocular antibiotic delivery with strong potential to improve outcomes in bacterial keratitis — particularly in deep stromal infections where conventional eye drops often fail.
First-of-its-Kind
First-ever besifloxacin-eluting contact lens reported in scientific literature. Novel EC/RSPO polymer blend for besifloxacin delivery.
Comparable Physical Properties
Light transmittance and water content equivalent to commercial CLs. Safe, comfortable to wear.
Sustained 24h+ Drug Release
Up to 15.4× more total drug delivered vs conventional soaked lenses. Clinically relevant drug levels maintained throughout wear.
21.6× Higher Cmax
Peak besifloxacin concentration in aqueous humor 21.6-fold greater than hourly eye drops. 18.3× enhanced AUC — dramatically improved bioavailability.
Proven Stability
Maintains drug integrity and lens properties after gamma irradiation sterilization and 2 years of dry storage at 4°C.
Safe & Biocompatible
No cytotoxicity (ISO 10993-5), no ocular irritation (Draize test), no corneal damage (fluorescein staining) — confirmed in human cells and rabbit model.
Funded by NIH R01EY026640 & P30EY003790 | International Journal of Pharmaceutics: X, 11 (2026) 100511
Critical Appraisal of the Study
📋 JOURNAL CLUB
STRENGTHS
LIMITATIONS / WEAKNESSES
Novelty
First BXF-eluting CL; first EC/RSPO combination for BXF delivery
Rigorous characterization
OCT, HPLC, spectrophotometry, gravimetry, LC-MS/MS extensively used
Comparative design
Head-to-head vs clinical standard (hourly eye drops) AND soaked commercial CLs
Clinically relevant PK outcomes
Aqueous humor as surrogate for corneal levels, 3Rs-compliant non-terminal design
Comprehensive stability
UV, autoclaving, gamma irradiation, and 2-year functional storage tested
Validated safety protocols
ISO 10993-5 cytotoxicity, Draize test (OECD), in vivo fluorescein staining
Quantitative antimicrobial confirmation
ZOI directly correlated to concentration via standard curve to validate bioactivity
Clear therapeutic thresholds cited
MIC₉₀ for S. aureus, Gram−, MRSA included to establish clinical translatability
Small animal model constraint
n = 3–6 per timepoint; forced non-parametric statistics due to limited sample size
No in vivo infection model
Efficacy in an actual bacterial keratitis infection model was not demonstrated
No hydrated storage testing
Current form requires pre-use hydration posing a clinical logistics challenge
E. coli vs P. aeruginosa evaluated
A more relevant contact lens-related keratitis pathogen was not directly tested
No long-term in vivo wear study
In vivo evaluation was stringently limited to a ≤48h wear time envelope
Rabbit vs human pharmacokinetics
Translational gap remains; robust human PK data needed to map clinical reality
AUC for eye drops overestimated
Acknowledged by authors: makes BXF-CL comparative outcome overly conservative
Single formulation design
Only methafilcon hydrogel explored; parallel alternative lens materials not compared
OVERALL: Well-designed translational proof-of-concept study with honest discussion of limitations. Strong foundation for clinical translation.
Source: Critical analysis of Sections 3–4 | ISO standards | OECD guidelines
References
Full reference list available in Kuang et al., 2026 — Int. J. Pharmaceutics: X 11, 100511
Kuang L et al. (2026). Besifloxacin-eluting contact lens with sustained drug delivery. Int J Pharm X, 11, 100511.
McDonald EM et al. (2014). Topical antibiotics for bacterial keratitis. Br J Ophthalmol, 98, 1470–1477.
Ung L et al. (2019). Microbial keratitis: global burden & antimicrobial resistance. Surv Ophthalmol, 64, 255–271.
Ross AE et al. (2019). Topical sustained drug delivery to retina with drug-eluting CL. Biomaterials, 217, 119285.
Peng C et al. (2022). Bibliometric analysis of ocular drug delivery 2001–2020. J Control Release, 345, 625–645.
Jumelle C et al. (2020). Advances & limitations of eye drop delivery systems. J Control Release, 321, 1–22.
Comstock TL et al. (2010). Besifloxacin: novel anti-infective for bacterial conjunctivitis. Clin Ophthalmol, 4, 215–225.
dos Santos GA et al. (2020). Besifloxacin liposomes for improved ocular delivery. Sci Rep, 10, 1–18.
Proksch JW et al. (2009). Ocular pharmacokinetics of besifloxacin. J Ocul Pharmacol Ther, 25, 335–344.
Bengani LC et al. (2020). Steroid-eluting contact lenses for ocular inflammation. Acta Biomater, 116, 149–161.
Galante R et al. (2018). Drug-eluting silicone hydrogel: impact of sterilization. Colloids Surf B, 161, 537–546.
Shah VP et al. (1997). FDA guidance: dissolution testing of immediate release solid dosage forms. Dissolution Technol, 4, 15–22.
Mah FS & Sanfilippo CM (2016). Besifloxacin: efficacy and safety. Ophthalmol Ther, 5, 1–20.
Stone JL et al. (2009). Objective evaluation of eyedrop instillation in glaucoma patients. Arch Ophthalmol, 127, 732–736.
Wilhelmus KR (2001). The Draize eye test. Surv Ophthalmol, 45, 493–515.
Ciolino JB et al. (2014). In vivo drug-eluting CL for glaucoma treatment. Biomaterials, 35, 432–439.
Lanier OL et al. (2020). Commercialization challenges for drug eluting contact lenses. Expert Opin Drug Deliv, 17, 1133–1149.
Polat HK et al. (2022). Novel drug delivery systems to improve keratitis treatment. J Ocul Pharmacol Ther, 38, 376–395.
Thank You
Questions & Discussion
For evaluators and audience:
How does the EC/RSPO polymer blend mechanistically contribute to controlled release — and what are the pharmacokinetic implications of the 8h Tmax compared to conventional eye drops?
The study compares BXF-CL to hourly eye drops — is this a clinically fair comparison? What are the statistical limitations of the small sample size?
How might the transition from rabbit to human pharmacokinetics change the observed 21.6-fold Cmax enhancement?
21.6×
Higher Peak Drug Level vs. Hourly Eye Drops
2 Years
Proven Shelf Life
24h
Sustained Therapeutic Release
Kuang L, Boychev N, Chen L et al. Besifloxacin-Eluting Contact Lens with Sustained Drug Delivery and Enhanced Bioavailability. Int. J. Pharmaceutics: X, 11 (2026) 100511. DOI: 10.1016/j.ijpx.2026.100511
NIH Funded: R01EY026640 | P30EY003790
- bacterial-keratitis
- ophthalmology
- drug-delivery-systems
- contact-lenses
- pharmacokinetics
- besifloxacin
- biomedical-engineering