Pathogenesis of Emphysema: Alveolar Destruction Mechanisms
Explore the mechanisms of emphysema, including protease imbalance, oxidative stress, and chronic inflammation in this comprehensive medical presentation.
Pathogenesis of Emphysema
Mechanisms of Alveolar Destruction & Disease Progression
Contents
A comprehensive journey through the mechanisms of emphysema.
What is Emphysema?
Definition & Disease Overview
Epidemiology & Risk Factors
Demographics, Smoking & Environmental Exposures
Types of Emphysema
Centriacinar, Panacinar & Paraseptal Classifications
Protease–Antiprotease Imbalance
Alpha-1 Antitrypsin Deficiency & Enzymatic Destruction
Oxidative Stress Mechanisms
Exogenous Toxins & Reactive Oxygen Species
Chronic Inflammation Cascade
Macrophages, Neutrophils & Cytokine Signaling
Alveolar Cell Death & Apoptosis
Loss of Epithelial & Endothelial Integrity
Impaired Lung Repair & Remodeling
Defective Extracellular Matrix Restoration
Integrated Pathogenic Model
Synthesis of Destructive Pathways
Clinical Manifestations & Complications
Symptomatology & Systemic Consequences
What is Emphysema?
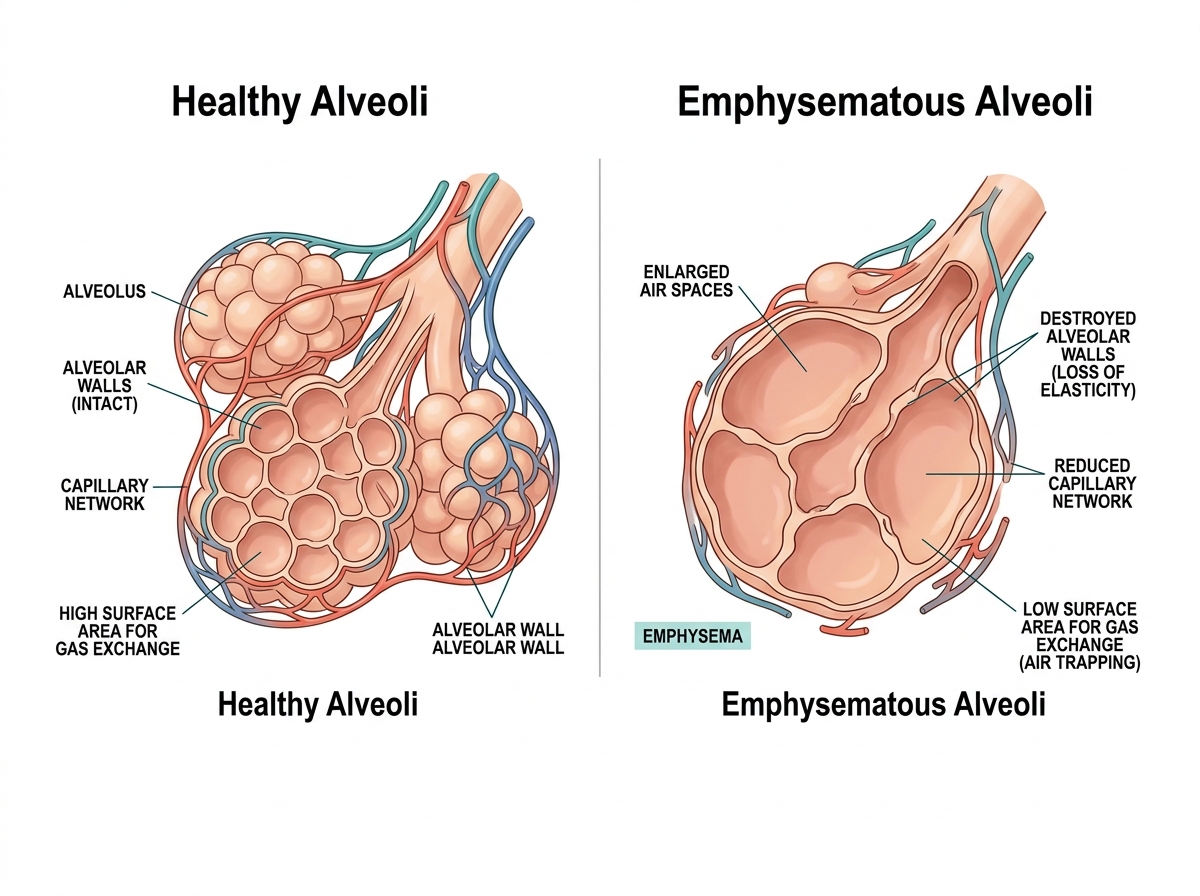
Emphysema is a chronic obstructive pulmonary disease (COPD) characterized by permanent, abnormal enlargement of the airspaces distal to the terminal bronchiole, accompanied by destruction of alveolar walls, without obvious fibrosis.
Permanent alveolar enlargement
Destruction of alveolar walls
Loss of lung elasticity & reduced gas exchange
Part of the COPD spectrum alongside chronic bronchitis
Normal vs. Emphysematous Alveoli
Epidemiology & Risk Factors
Global Burden
380M+
people affected worldwide
3rd
leading cause of death globally
10-15%
of smokers develop COPD/emphysema
↑ Age
Prevalence directly correlates with advancing age
Primary Risk Factors
Cigarette Smoking (80–90% of cases)
Air Pollution & Occupational Dust
Alpha-1 Antitrypsin Deficiency (1–2%)
Recurrent Pulmonary Infections
Low Birth Weight / Premature Birth
Aging
Alpha-1 Antitrypsin Deficiency
Autosomal codominant inheritance
PiZZ genotype: highest risk
Causes panacinar emphysema
Affects lower lobes predominantly
Gene therapy under active research
Types of Emphysema
Centriacinar
(Centrilobular)
Associated with cigarette smoking
Predominantly UPPER lobes
Affects respiratory bronchioles centrally
Most common type (95%)
Panacinar
(Panlobular)
Associated with Alpha-1 Antitrypsin deficiency
Predominantly LOWER lobes
Affects entire acinus uniformly
More severe gas exchange impairment
Paraseptal
(Distal Acinar)
Often seen in young adults
Adjacent to pleura and septa
Affects distal alveolar ducts & sacs
Can cause spontaneous pneumothorax
Irregular
(Scar Emphysema)
Associated with scarring / fibrosis
Irregular distribution throughout
Not linked to airway obstruction
Usually an incidental finding
04
The Protease–Antiprotease Imbalance
ESTABLISHED THEORY — 1960s, REMAINS CENTRAL TODAY
PROTEASES
— EXCESS —
Neutrophil Elastase
Matrix Metalloproteases
(MMP-1, 9, 12)
Macrophage Elastase
Cathepsins
(B, L, S)
ANTIPROTEASES
— DEPLETED —
Alpha-1 Antitrypsin (AAT)
PRIMARY INHIBITOR
SLPI
(Secretory Leukocyte Protease Inhibitor)
Elafin
TIMPs
CIGARETTE SMOKE
disrupts balance
CONSEQUENCE OF EXCESS
Elastin Degradation
Loss of alveolar wall integrity
GENETIC FACTOR
AAT Deficiency
Genetic predisposition to early emphysema
05
Oxidative Stress Mechanisms
Cigarette Smoke & Pollutants
↑ Reactive Oxygen Species (ROS) & RNS
↓ Antioxidant Depletion
Glutathione, Vitamin A & E
SOD, Catalase
Nrf2 activity reduced
OXIDANT–ANTIOXIDANT IMBALANCE
DNA Damage & Telomere Shortening
Antiprotease Inactivation
→ Compounds protease imbalance
HDAC2 Inactivation
→ Perpetuates pro-inflammatory gene expression
Apoptosis Activation
→ Alveolar cell death
Oxidative stress markers persist even in ex-smokers — indicating endogenous ongoing inflammation
Chronic Inflammation Cascade
Cigarette Smoke / Irritant Exposure
Elastin/Collagen fragments → Matrikines → Further inflammation amplification → Self-perpetuating cycle
Alveolar Cell Death & Apoptosis
APOPTOSIS
Programmed Cell Death
Activated by oxidative stress
Caspase cascade activation
Affects: Epithelial, Endothelial, Fibroblast cells
Releases DAMPs → amplifies inflammation
AUTOPHAGY
Cellular Self-Digestion
mTOR pathway disruption
Cigarette smoke activates
Mitochondrial dysfunction
NLRP3 inflammasome activation
NECROSIS
Uncontrolled Cell Death
Secondary to severe oxidative damage
Inflammatory cell infiltration
cGAS-STING pathway activation
DNA sensing mechanisms
VEGF — Critical Survival Factor
Abundantly expressed in healthy lungs
Required for endothelial cell survival
VEGF reduction → airspace enlargement (inflammation-independent)
Targeted in experimental therapies
Ceramide Pathway
Endogenous lipid mediator
Activated by cigarette smoke
Engages: apoptosis + protease imbalance + oxidative stress
Persists AFTER smoking cessation ⚠️
Loss of alveolar surface area reduces gas exchange capacity permanently
Impaired Lung Repair & Remodeling
Emphysema reflects not only destruction — but failure to repair
mTOR — Stress Sensor
Mammalian Target of Rapamycin
Critical sensor for stress response
Regulates alveolar maintenance programs
Rtp801 activated by cigarette smoke → inhibits mTOR → airspace enlargement
Alveolar Maintenance FAILS
Accelerated Lung Aging
Telomeres shorten in alveolar cells
Promotes cellular senescence
Increases susceptibility to exacerbations
Mitochondrial dysfunction co-occurs
Premature Aging of Lung Tissue
Tissue Remodeling Failure
Notch & Wnt pathways govern differentiation
Epithelial & mesenchymal cell fate
Combined Pulmonary Fibrosis & Emphysema (CPFE)
Emerging area of research (2024–2025)
Abnormal Tissue Remodeling
Once initiated, multiple reinforcing loops perpetuate progression — even without ongoing smoke exposure
Integrated Pathogenic Model
Four synergistic mechanisms driving alveolar destruction
Only ~10–15% of smokers develop emphysema — genetic susceptibility plays a key role
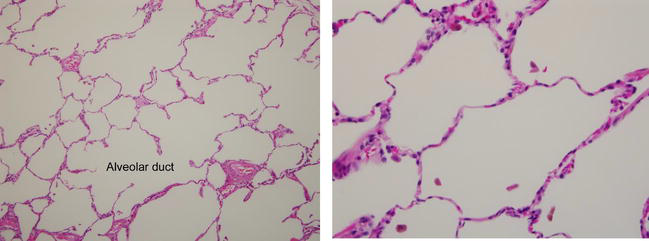
Histopathology
Microscopic features of emphysematous lung tissue
H&E stain: Normal (left) vs. Emphysematous (right) lung tissue
Clinical Manifestations & Complications
Classic Clinical Picture
Pink Puffer
Type A Emphysema
Barrel-shaped chest (hyperinflation)
Pursed-lip breathing
Prolonged expiration
Lean, thin patient
Relatively well-oxygenated until late
Accessory muscle use
Pulmonary Function Tests
↓ FEV1/FVC ratio (obstruction)
↓ FEV1 (airflow limitation)
↑ Total Lung Capacity (hyperinflation)
↑ Residual Volume (air trapping)
↓ DLCO (diffusion capacity — hallmark)
↓ Elastic recoil
Symptoms Progression
Insidious onset
Progressive exertional dyspnea
Chronic cough (less prominent)
Weight loss
Fatigue
Reduced exercise tolerance
Complications
Respiratory Failure
Cor Pulmonale
Spontaneous Pneumothorax
Recurrent Infections
Pulmonary Hypertension
Diagnosis & Imaging
Pulmonary Function
Imaging Findings
Laboratory & Other
Management & Treatment Strategies
14
Recent Research & Future Directions
Emerging discoveries reshaping our understanding of emphysema (2024–2026)
🤖
Machine Learning Discovery
Novel Emphysema Inflammatory Subgroup (EIS)
Identified via machine learning analysis (2024–2025)
Distinct inflammatory phenotype within emphysema spectrum
Highlights heterogeneity — not one disease, multiple subtypes
Implications for personalized medicine approaches
🔬
Emerging Syndrome
Combined Pulmonary Fibrosis & Emphysema (CPFE)
Coexistence of active fibrosis + alveolar destruction
Notch and Wnt signaling pathways implicated
More complex remodeling than classical models
Poorer prognosis than emphysema alone
💊
Therapeutic Target
Ceramide Pathway — Post-Cessation Progression
Disease progression independent of ongoing smoke
Ceramide activation persists after smoking cessation
Potential therapeutic target
Explains why some patients worsen after quitting
🧬
Novel Target 2025
NLRP3 Inflammasome & cGAS-STING
Mitochondrial dysfunction activates NLRP3
DNA sensing via cGAS-STING pathway
Novel anti-inflammatory targets
Small molecule inhibitors in trials
Understanding the precise molecular mechanisms opens doors to targeted therapies beyond bronchodilation
Key Takeaways
What every medical student should remember
Emphysema = permanent airspace enlargement + alveolar wall destruction WITHOUT fibrosis — the definition to remember
4 Core Mechanisms: Protease–Antiprotease Imbalance + Oxidative Stress + Chronic Inflammation + Cell Death/Repair Failure — all synergistic
Smoking is #1 cause (80–90%) — but only 10–15% of smokers develop it — genetic susceptibility matters
Alpha-1 Antitrypsin Deficiency = most important genetic cause — causes panacinar emphysema of lower lobes
↓ DLCO is the hallmark PFT finding — distinguishes emphysema from chronic bronchitis
Smoking cessation is the only intervention proven to slow disease progression — prevention is paramount
Review Complete — 15 Topics Covered
Thank You
Questions & Discussion
Every breath reminds us of the intricate machinery we must protect. Understanding its failure is the first step to healing.
— Pulmonary Pathology
Global Initiative for Chronic Obstructive Lung Disease (GOLD) 2024
Barnes PJ. New concepts in COPD. Annu Rev Med. 2023
Stockley RA et al. Emphysema Mechanisms. Thorax. 2024
Pathogenesis of Emphysema
Presented by [Your Name]
2026 | Medical Student Presentation
- emphysema
- pathogenesis
- pulmonary-pathology
- medical-education
- copd
- alveolar-destruction
- respiratory-disease