GCSE Foundation Chemistry: Reactivity, Redox & Synthesis
Master GCSE Chemistry with this guide on the reactivity series, redox reactions, extraction of metals, and salt synthesis methods for Edexcel Foundation.
GCSE Foundation Chemistry
Reactivity Series | Redox Reactions | Synthesis
Edexcel | 2 Hour Lesson
Lesson Overview
Part 1: Reactivity Series
The reactivity series order
Displacement reactions
Reactions with water & acids
Extracting metals
Part 2: Redox Reactions
Oxidation & reduction
OIL RIG mnemonic
Electron transfer
Identifying redox reactions
Part 3: Synthesis
Making salts
Neutralisation reactions
Salt preparation methods
Core practical
⏱ 2 Hour Lesson | Edexcel GCSE Foundation Chemistry
Part 1
Reactivity Series
Ordering metals, displacement reactions & extracting metals from ores
The Reactivity Series
Reaction Key
React with cold water
React with steam only
React with dilute acid
Very unreactive
"Please Send Little Cats Meowing About Zebras In The Lovely Calm Seas Going"
<div style="display: flex; align-items: center; justify-content: space-between; background-color: #0b3d54; padding: 0 25px; border-radius: 8px; flex: 1;"> <div style="display: flex; align-items: baseline;"> <span style="color: #00d2ff; font-weight: 700; font-size: 22px; width: 45px;">1.</span> <span style="color: #ffffff; font-weight: 600; font-size: 26px;">Potassium (K)</span> <span style="color: #a0aec0; font-weight: 400; font-size: 20px; margin-left: 15px;">- most reactive</span> </div> <span style="font-size: 24px;">🔵</span> </div> <div style="display: flex; align-items: center; justify-content: space-between; background-color: #111a2f; padding: 0 25px; border-radius: 8px; flex: 1;"> <div style="display: flex; align-items: baseline;"> <span style="color: #00d2ff; font-weight: 700; font-size: 22px; width: 45px;">2.</span> <span style="color: #ffffff; font-weight: 600; font-size: 26px;">Sodium (Na)</span> </div> <span style="font-size: 24px;">🔵</span> </div> <div style="display: flex; align-items: center; justify-content: space-between; background-color: #0b3d54; padding: 0 25px; border-radius: 8px; flex: 1;"> <div style="display: flex; align-items: baseline;"> <span style="color: #00d2ff; font-weight: 700; font-size: 22px; width: 45px;">3.</span> <span style="color: #ffffff; font-weight: 600; font-size: 26px;">Lithium (Li)</span> </div> <span style="font-size: 24px;">🔵</span> </div> <div style="display: flex; align-items: center; justify-content: space-between; background-color: #111a2f; padding: 0 25px; border-radius: 8px; flex: 1;"> <div style="display: flex; align-items: baseline;"> <span style="color: #00d2ff; font-weight: 700; font-size: 22px; width: 45px;">4.</span> <span style="color: #ffffff; font-weight: 600; font-size: 26px;">Calcium (Ca)</span> </div> <span style="font-size: 24px;">🔵</span> </div> <div style="display: flex; align-items: center; justify-content: space-between; background-color: #0b3d54; padding: 0 25px; border-radius: 8px; flex: 1;"> <div style="display: flex; align-items: baseline;"> <span style="color: #00d2ff; font-weight: 700; font-size: 22px; width: 45px;">5.</span> <span style="color: #ffffff; font-weight: 600; font-size: 26px;">Magnesium (Mg)</span> </div> <span style="font-size: 24px;">🟡</span> </div> <div style="display: flex; align-items: center; justify-content: space-between; background-color: #111a2f; padding: 0 25px; border-radius: 8px; flex: 1;"> <div style="display: flex; align-items: baseline;"> <span style="color: #00d2ff; font-weight: 700; font-size: 22px; width: 45px;">6.</span> <span style="color: #ffffff; font-weight: 600; font-size: 26px;">Aluminium (Al)</span> </div> <span style="font-size: 24px;">🟡</span> </div> <div style="display: flex; align-items: center; justify-content: space-between; background-color: #0b3d54; padding: 0 25px; border-radius: 8px; flex: 1;"> <div style="display: flex; align-items: baseline;"> <span style="color: #00d2ff; font-weight: 700; font-size: 22px; width: 45px;">7.</span> <span style="color: #ffffff; font-weight: 600; font-size: 26px;">Zinc (Zn)</span> </div> <span style="font-size: 24px;">🟡</span> </div> <div style="display: flex; align-items: center; justify-content: space-between; background-color: #111a2f; padding: 0 25px; border-radius: 8px; flex: 1;"> <div style="display: flex; align-items: baseline;"> <span style="color: #00d2ff; font-weight: 700; font-size: 22px; width: 45px;">8.</span> <span style="color: #ffffff; font-weight: 600; font-size: 26px;">Iron (Fe)</span> </div> <span style="font-size: 24px;">🟡</span> </div> <div style="display: flex; align-items: center; justify-content: space-between; background-color: #0b3d54; padding: 0 25px; border-radius: 8px; flex: 1;"> <div style="display: flex; align-items: baseline;"> <span style="color: #00d2ff; font-weight: 700; font-size: 22px; width: 45px;">9.</span> <span style="color: #ffffff; font-weight: 600; font-size: 26px;">Tin (Sn)</span> </div> <span style="font-size: 24px;">🔴</span> </div> <div style="display: flex; align-items: center; justify-content: space-between; background-color: #111a2f; padding: 0 25px; border-radius: 8px; flex: 1;"> <div style="display: flex; align-items: baseline;"> <span style="color: #00d2ff; font-weight: 700; font-size: 22px; width: 45px;">10.</span> <span style="color: #ffffff; font-weight: 600; font-size: 26px;">Lead (Pb)</span> </div> <span style="font-size: 24px;">🔴</span> </div> <div style="display: flex; align-items: center; justify-content: space-between; background-color: rgba(255, 215, 0, 0.12); padding: 0 25px; border-radius: 8px; flex: 1; border: 2px dashed rgba(255, 215, 0, 0.6);"> <div style="display: flex; align-items: baseline;"> <span style="color: #ffd700; font-weight: 700; font-size: 22px; width: 45px;">11.</span> <span style="color: #ffd700; font-weight: 800; font-size: 26px; letter-spacing: 0.5px;">HYDROGEN (H)</span> </div> <span style="color: #ffd700; font-size: 16px; font-weight: 600; font-style: italic;">non-metal</span> </div> <div style="display: flex; align-items: center; justify-content: space-between; background-color: #111a2f; padding: 0 25px; border-radius: 8px; flex: 1;"> <div style="display: flex; align-items: baseline;"> <span style="color: #00d2ff; font-weight: 700; font-size: 22px; width: 45px;">12.</span> <span style="color: #ffffff; font-weight: 600; font-size: 26px;">Copper (Cu)</span> </div> <span style="font-size: 24px;">⚫</span> </div> <div style="display: flex; align-items: center; justify-content: space-between; background-color: #0b3d54; padding: 0 25px; border-radius: 8px; flex: 1;"> <div style="display: flex; align-items: baseline;"> <span style="color: #00d2ff; font-weight: 700; font-size: 22px; width: 45px;">13.</span> <span style="color: #ffffff; font-weight: 600; font-size: 26px;">Silver (Ag)</span> </div> <span style="font-size: 24px;">⚫</span> </div> <div style="display: flex; align-items: center; justify-content: space-between; background-color: #111a2f; padding: 0 25px; border-radius: 8px; flex: 1;"> <div style="display: flex; align-items: baseline;"> <span style="color: #00d2ff; font-weight: 700; font-size: 22px; width: 45px;">14.</span> <span style="color: #ffffff; font-weight: 600; font-size: 26px;">Gold (Au)</span> <span style="color: #a0aec0; font-weight: 400; font-size: 20px; margin-left: 15px;">- least reactive</span> </div> <span style="font-size: 24px;">⚫</span> </div>
Reactions with Water and Acids
Reactions with Water
Reactions with Dilute Acids
2Na(s) + 2H₂O(l) → 2NaOH(aq) + H₂(g)
Mg(s) + 2HCl(aq) → MgCl₂(aq) + H₂(g)
The gas produced is always HYDROGEN — tested with a lit splint (squeaky pop!)
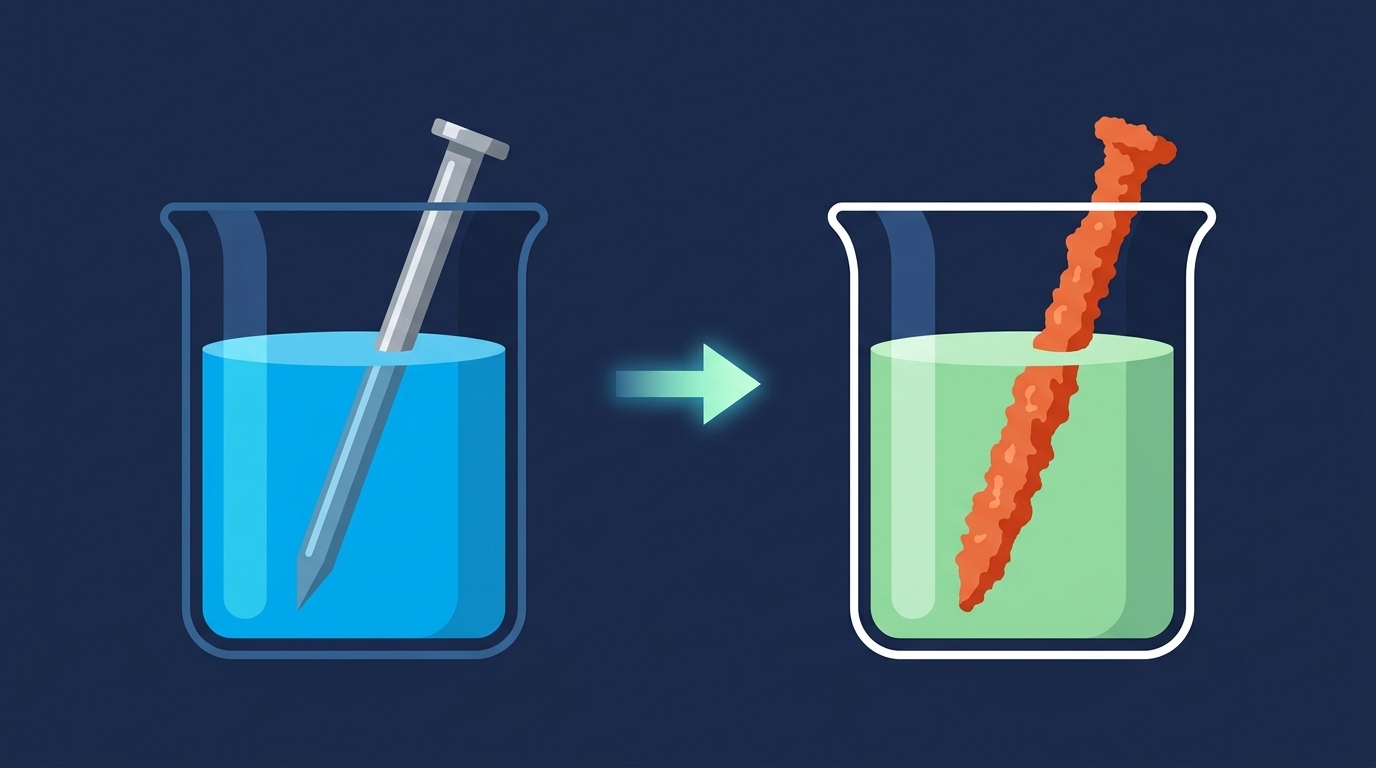
Displacement Reactions
A
MORE
reactive metal will
DISPLACE
a
LESS
reactive metal from its salt solution.
Reaction Occurs
Zn + CuSO₄ → ZnSO₄ + Cu
Blue solution goes colourless, orange/red copper metal forms
Reaction Occurs
Fe + CuSO₄ → FeSO₄ + Cu
Blue solution fades, copper deposits on iron
No Reaction
Cu + ZnSO₄ → No reaction
No change — copper is LESS reactive than zinc
More reactive =
pushes the less reactive metal
OUT
of its compound.
Extracting Metals from Ores
How a metal is extracted depends on its position in the reactivity series.
Very Reactive Metals
Potassium, Sodium, Lithium, Calcium, Magnesium, Aluminium
ELECTROLYSIS
Too reactive to be reduced by carbon. Requires large amounts of electricity.
Moderately Reactive Metals
Zinc, Iron, Tin, Lead
REDUCTION WITH CARBON
ZnO + C → Zn + CO₂
Carbon is more reactive than these metals so it displaces them.
Unreactive Metals
Copper, Silver, Gold
FOUND NATIVE or SIMPLE HEATING
So unreactive they exist as pure metals in the ground.
Aluminium is extracted by electrolysis despite being common — its oxide is very stable.
📝 EXAM QUESTIONS — Reactivity Series
Place the following metals in order of reactivity, starting with the most reactive: Copper, Magnesium, Zinc, Gold
Iron is added to copper sulfate solution. Describe what you would observe and explain why this reaction occurs.
A student investigates the reactions of four metals (A, B, C, D) with dilute hydrochloric acid. Metal A fizzes vigorously, metal B fizzes slowly, metal C shows no reaction, and metal D reacts explosively.
Place metals A, B, C, D in order of reactivity, most reactive first.
Predict what would happen if metal A was added to a solution of metal B sulfate.
Explain why gold is found as a pure metal in the ground, whereas aluminium is not.
⏱ Suggested time: 10 minutes | Put your pen down when done!
ATTEMPT THESE BEFORE LOOKING AT THE NEXT SLIDE →
✅ ANSWERS — Reactivity Series
A1 [2 marks]
Most reactive → Least reactive: Magnesium, Zinc, Copper, Gold. (1 mark for correct position of Magnesium/Gold; 1 mark for correct full order)
A2 [3 marks]
Observation: The blue colour of the solution fades/goes pale green. Orange/red solid (copper) forms on the iron. (2 marks for observations) Explanation: Iron is more reactive than copper, so iron displaces copper from the solution. (1 mark)
A3 [4 marks]
(a) Most reactive first: D, A, B, C — D reacts explosively (most reactive), A vigorously, B slowly, C no reaction. (2 marks for correct full order) (b) Metal A would displace metal B from its sulfate solution because A is more reactive than B. The solution would change colour and a solid deposit of metal B would form. (2 marks)
A4 [2 marks]
Gold is very unreactive / low in the reactivity series (1 mark), so it does not react with oxygen or other compounds in the ground and exists in its pure/native form. (1 mark)
💡 Examiner Tip:
Always use the word DISPLACE in displacement reaction answers!
📝 EXAM QUESTIONS — Reactivity Series (Set 2)
[1 mark]
Which metal would react most vigorously with cold water?<br><br><div style="display: grid; grid-template-columns: 1fr 1fr; gap: 20px; margin-top: 5px; padding-left: 10px;"><div><b style='color:#ffb347; margin-right: 10px;'>A)</b> Copper</div><div><b style='color:#ffb347; margin-right: 10px;'>B)</b> Iron</div><div><b style='color:#ffb347; margin-right: 10px;'>C)</b> Potassium</div><div><b style='color:#ffb347; margin-right: 10px;'>D)</b> Zinc</div></div>
[3 marks]
Lead oxide (PbO) can be reduced using carbon. Write a word equation for this reaction and explain why carbon can be used to extract lead but cannot be used to extract aluminium.
[4 marks]
A student places a piece of magnesium into zinc sulfate solution and a separate piece of magnesium into copper sulfate solution.<br><br><div style='display: flex; gap: 12px; margin-bottom: 16px;'><b style='color:#ffb347; white-space: nowrap;'>(a)</b><span style='flex-grow: 1;'>Write a word equation for the reaction between magnesium and copper sulfate.</span><span style='color:#a0aec0; font-size:22px; white-space: nowrap;'>[1]</span></div><div style='display: flex; gap: 12px; margin-bottom: 16px;'><b style='color:#ffb347; white-space: nowrap;'>(b)</b><span style='flex-grow: 1;'>Write a balanced symbol equation for the reaction between magnesium and copper sulfate.</span><span style='color:#a0aec0; font-size:22px; white-space: nowrap;'>[2]</span></div><div style='display: flex; gap: 12px;'><b style='color:#ffb347; white-space: nowrap;'>(c)</b><span style='flex-grow: 1;'>Which reaction would you expect to be more vigorous? Explain your answer.</span><span style='color:#a0aec0; font-size:22px; white-space: nowrap;'>[1]</span></div>
[2 marks]
Explain why iron objects left outdoors rust, but gold objects do not.
⏱ Suggested time: 10 minutes | ATTEMPT BEFORE TURNING OVER →
ANSWERS — Reactivity Series (Set 2)
Examiner Tip:
For extraction questions, always state which is MORE reactive — the metal or carbon.
A1 [1 mark]
A2 [3 marks]
A3 [4 marks]
A4 [2 marks]
Part 2
Redox Reactions
Oxidation, Reduction & Electron Transfer
OIL RIG
Oxidation and Reduction
OIL — Oxidation Is Loss
Oxidation = <b>LOSS</b> of electrons
The oxidised species <b>LOSES</b> electrons
Oxidation = GAIN of oxygen
Mg → Mg²⁺ + 2e⁻
(magnesium is OXIDISED)
RIG — Reduction Is Gain
Reduction = <b>GAIN</b> of electrons
The reduced species <b>GAINS</b> electrons
Reduction = LOSS of oxygen
Cu²⁺ + 2e⁻ → Cu
(copper ion is REDUCED)
REDOX reactions <b>ALWAYS</b> happen together — if something is oxidised, something else <b>MUST</b> be reduced at the same time!
Redox in Displacement Reactions
Overall Reaction:
Zn(s) + CuSO₄(aq) → ZnSO₄(aq) + Cu(s)
Oxidation Half-Equation
Zn → Zn²⁺ + 2e⁻
Zinc is OXIDISED
Zinc LOSES 2 electrons
Reduction Half-Equation
Cu²⁺ + 2e⁻ → Cu
Copper ion is REDUCED
Copper ion GAINS 2 electrons
The electrons lost by zinc are GAINED by copper ions.
Zinc = REDUCING AGENT
(it gets oxidised, causes reduction of copper)
Copper = OXIDISING AGENT
(it gets reduced, causes oxidation of zinc)
Colour change
Blue solution → Colourless
Orange copper solid forms
Redox in Terms of Oxygen
Oxidation = Gain of Oxygen
Reduction = Loss of Oxygen
2Mg + O₂ → 2MgO
Magnesium is <strong style="color:#00e5ff;">OXIDISED</strong> (gains oxygen)
C + O₂ → CO₂
Carbon is <strong style="color:#00e5ff;">OXIDISED</strong>
Fe + S → FeS
Iron is <strong style="color:#00e5ff;">OXIDISED</strong> (gains non-metal)
CuO + H₂ → Cu + H₂O
Copper oxide is <strong style="color:#d500f9;">REDUCED</strong> (loses oxygen)
PbO + C → Pb + CO₂
Lead oxide is <strong style="color:#d500f9;">REDUCED</strong>
Fe₂O₃ + 3CO → 2Fe + 3CO₂
Iron ore is <strong style="color:#d500f9;">REDUCED</strong> in a blast furnace
CuO + Mg → Cu + MgO
CuO is <strong style="color:#d500f9; font-weight:800;">REDUCED</strong> (loses oxygen)
Mg is <strong style="color:#00e5ff; font-weight:800;">OXIDISED</strong> (gains oxygen)
THIS IS A REDOX REACTION
<span style="color:#ffeb3b; font-size:28px; margin-right:8px; vertical-align:middle;">💡</span><span style="vertical-align:middle;">In the extraction of metals, the metal oxide is always <strong style="color:#d500f9;">REDUCED</strong> and the reducing agent (carbon/CO) is <strong style="color:#00e5ff;">OXIDISED</strong>.</span>
📝 EXAM QUESTIONS — Redox Reactions
1 Mark
<div style="margin-bottom: 15px;">In the reaction:</div> <div style="font-size: 28px; font-weight: bold; font-family: 'Consolas', 'Courier New', monospace; background: rgba(0,0,0,0.3); padding: 15px; text-align: center; margin-bottom: 25px; border-radius: 8px;">Fe + CuSO₄ → FeSO₄ + Cu</div> <div style="margin-bottom: 20px; font-weight: 600;">Which species is oxidised?</div> <div style="display: grid; grid-template-columns: 1fr 1fr; gap: 15px;"> <div style="background: rgba(255,255,255,0.08); padding: 12px 20px; border-radius: 8px; border: 1px solid rgba(255,255,255,0.1);"><span style="color: #00d2ff; font-weight: bold; margin-right: 15px;">A)</span> Fe</div> <div style="background: rgba(255,255,255,0.08); padding: 12px 20px; border-radius: 8px; border: 1px solid rgba(255,255,255,0.1);"><span style="color: #00d2ff; font-weight: bold; margin-right: 15px;">B)</span> Cu²⁺</div> <div style="background: rgba(255,255,255,0.08); padding: 12px 20px; border-radius: 8px; border: 1px solid rgba(255,255,255,0.1);"><span style="color: #00d2ff; font-weight: bold; margin-right: 15px;">C)</span> SO₄²⁻</div> <div style="background: rgba(255,255,255,0.08); padding: 12px 20px; border-radius: 8px; border: 1px solid rgba(255,255,255,0.1);"><span style="color: #00d2ff; font-weight: bold; margin-right: 15px;">D)</span> Fe²⁺</div> </div>
3 Marks
<div style="margin-bottom: 20px;">Zinc is added to silver nitrate solution.</div> <div style="display: flex; gap: 15px; margin-bottom: 12px;"><span style="color: #00d2ff; font-weight: 700;">(a)</span> <span style="flex-grow: 1;">Write a word equation for this reaction.</span> <span style="font-size: 20px; color: rgba(0, 210, 255, 0.7);">[1]</span></div> <div style="display: flex; gap: 15px; margin-bottom: 12px;"><span style="color: #00d2ff; font-weight: 700;">(b)</span> <span style="flex-grow: 1;">Identify which substance is oxidised and which is reduced.</span> <span style="font-size: 20px; color: rgba(0, 210, 255, 0.7);">[1]</span></div> <div style="display: flex; gap: 15px; margin-bottom: 12px;"><span style="color: #00d2ff; font-weight: 700;">(c)</span> <span style="flex-grow: 1;">Explain what happens to electrons during this reaction.</span> <span style="font-size: 20px; color: rgba(0, 210, 255, 0.7);">[1]</span></div>
4 Marks
<div style="margin-bottom: 10px;">The following reaction occurs in a blast furnace:</div> <div style="font-size: 26px; font-weight: bold; font-family: 'Consolas', 'Courier New', monospace; background: rgba(0,0,0,0.3); padding: 12px; text-align: center; margin-bottom: 15px; border-radius: 8px;">Fe₂O₃ + 3CO → 2Fe + 3CO₂</div> <div style="display: flex; gap: 12px; margin-bottom: 8px;"><span style="color: #00d2ff; font-weight: 700;">(a)</span> <span style="flex-grow: 1;">Identify which substance is oxidised.</span> <span style="font-size: 18px; color: rgba(0, 210, 255, 0.7);">[1]</span></div> <div style="display: flex; gap: 12px; margin-bottom: 8px;"><span style="color: #00d2ff; font-weight: 700;">(b)</span> <span style="flex-grow: 1;">Identify which substance is reduced.</span> <span style="font-size: 18px; color: rgba(0, 210, 255, 0.7);">[1]</span></div> <div style="display: flex; gap: 12px; margin-bottom: 8px;"><span style="color: #00d2ff; font-weight: 700;">(c)</span> <span style="flex-grow: 1;">Explain, in terms of oxygen, why iron oxide is reduced.</span> <span style="font-size: 18px; color: rgba(0, 210, 255, 0.7);">[1]</span></div> <div style="display: flex; gap: 12px;"><span style="color: #00d2ff; font-weight: 700;">(d)</span> <span style="flex-grow: 1;">What is the role of carbon monoxide in this reaction?</span> <span style="font-size: 18px; color: rgba(0, 210, 255, 0.7);">[1]</span></div>
2 Marks
<div style="margin-bottom: 25px; font-size: 28px; line-height: 1.5;">Define the term <strong>'redox reaction'</strong>.</div> <div style="background: rgba(245, 158, 11, 0.1); border-left: 4px solid #f59e0b; padding: 20px 25px; border-radius: 0 8px 8px 0; font-style: italic;"> Use the terms <span style="color: #f59e0b; font-weight: 700; font-style: normal;">oxidation</span> and <span style="color: #f59e0b; font-weight: 700; font-style: normal;">reduction</span> in your answer. </div>
⏱ Suggested time: 10 minutes | ATTEMPT BEFORE TURNING OVER →
✅ ANSWERS — Redox Reactions
A1 [1 mark]
A) Fe ✓ — Iron loses electrons (Fe → Fe²⁺ + 2e⁻) so it is <strong style="color: #ffffff;">OXIDISED</strong>.<br><br>Remember OIL — Oxidation Is Loss of electrons.
A2 [3 marks]
<span style="color: #00e676; font-weight: 600;">(a)</span> Zinc + silver nitrate → zinc nitrate + silver (1 mark)<br><br><span style="color: #00e676; font-weight: 600;">(b)</span> Zinc is oxidised; Silver ions (Ag⁺) are reduced. (1 mark)<br><br><span style="color: #00e676; font-weight: 600;">(c)</span> Zinc atoms <strong style="color: #ffffff;">LOSE</strong> electrons (Zn → Zn²⁺ + 2e⁻). These electrons are <strong style="color: #ffffff;">GAINED</strong> by silver ions (Ag⁺ + e⁻ → Ag). (1 mark)
A3 [4 marks]
<span style="color: #00e676; font-weight: 600;">(a)</span> Carbon monoxide (CO) is oxidised — it gains oxygen to form CO₂. (1 mark)<br><br><span style="color: #00e676; font-weight: 600;">(b)</span> Iron oxide (Fe₂O₃) is reduced. (1 mark)<br><br><span style="color: #00e676; font-weight: 600;">(c)</span> Iron oxide <strong style="color: #ffffff;">LOSES</strong> oxygen — it goes from Fe₂O₃ to Fe (pure iron). Reduction = loss of oxygen. (1 mark)<br><br><span style="color: #00e676; font-weight: 600;">(d)</span> Carbon monoxide acts as the <strong style="color: #ffffff;">REDUCING AGENT</strong> — it removes oxygen from the iron oxide. (1 mark)
A4 [2 marks]
A redox reaction is a reaction in which oxidation and reduction occur simultaneously <span style="color: #8bb1d6;">(1 mark)</span>.<br><br>One species loses electrons/gains oxygen (oxidation) while another species gains electrons/loses oxygen (reduction) at the same time. <span style="color: #8bb1d6;">(1 mark)</span>
💡 Examiner Tip: Always name BOTH the oxidised AND reduced species — half marks are common if you only name one!
📝 EXAM QUESTIONS — Redox Reactions (Set 2)
In the reaction: 2H₂ + O₂ → 2H₂O. (a) Is hydrogen oxidised or reduced? [1 mark] (b) Is oxygen oxidised or reduced? [1 mark]
A student says: 'In a displacement reaction, the more reactive metal is always oxidised.' Explain whether the student is correct, using the example of magnesium reacting with iron sulfate solution.
Copper oxide is heated with hydrogen gas: CuO + H₂ → Cu + H₂O. (a) Write down what is oxidised in this reaction. [1 mark] (b) Write down what is reduced in this reaction. [1 mark] (c) In terms of electrons, explain the process of reduction. [1 mark] (d) What term do we use for a substance that causes the reduction of another substance? [1 mark]
Explain the meaning of the term 'reducing agent'. Give an example from a reaction you have studied.
✅ ANSWERS — Redox Reactions (Set 2)
A1
[2 marks]
<div style='margin-bottom: 12px;'>(a) Hydrogen is <span style='color: #ffffff; font-weight: 700;'>OXIDISED</span> — it gains oxygen to form water. <span style='color: #00d2ff; font-weight: 700;'>(1 mark)</span></div><div style='margin-bottom: 12px;'>(b) Oxygen is <span style='color: #ffffff; font-weight: 700;'>REDUCED</span> — it gains electrons / is part of the reduction process. <span style='color: #00d2ff; font-weight: 700;'>(1 mark)</span></div><div style='color: #94a3b8; font-size: 20px; font-style: italic; background: rgba(0,0,0,0.25); padding: 12px 18px; border-left: 4px solid #64748b; border-radius: 6px; margin-top: 15px;'>NOTE: At foundation, accept 'oxygen causes oxidation of hydrogen'.</div>
A2
[3 marks]
<div style='margin-bottom: 14px;'>The student is <span style='color: #4ade80; font-weight: 700;'>CORRECT</span> <span style='color: #00d2ff; font-weight: 700;'>(1 mark)</span>. <br>In the reaction: <span style='color: #ffffff; font-weight: 600;'>Mg + FeSO₄ → MgSO₄ + Fe</span>.</div><div style='margin-bottom: 14px;'>Magnesium (more reactive) is <span style='color: #ffffff; font-weight: 700;'>OXIDISED</span>: Mg → Mg²⁺ + 2e⁻ — it loses electrons. <span style='color: #00d2ff; font-weight: 700;'>(1 mark)</span></div><div>Iron ions (Fe²⁺) are <span style='color: #ffffff; font-weight: 700;'>REDUCED</span>: Fe²⁺ + 2e⁻ → Fe — they gain electrons. The more reactive metal always loses electrons (is oxidised). <span style='color: #00d2ff; font-weight: 700;'>(1 mark)</span></div>
A3
[4 marks]
<div style='margin-bottom: 14px;'>(a) Hydrogen (H₂) is oxidised — it gains oxygen. <span style='color: #00d2ff; font-weight: 700;'>(1 mark)</span></div><div style='margin-bottom: 14px;'>(b) Copper oxide (CuO) is reduced — it loses oxygen. <span style='color: #00d2ff; font-weight: 700;'>(1 mark)</span></div><div style='margin-bottom: 14px;'>(c) Reduction involves the <span style='color: #ffffff; font-weight: 700;'>GAIN</span> of electrons. In this reaction, copper ions gain electrons to become copper atoms: <span style='color: #ffffff; font-weight: 600;'>Cu²⁺ + 2e⁻ → Cu</span>. <span style='color: #00d2ff; font-weight: 700;'>(1 mark)</span></div><div>(d) A <span style='color: #ffffff; font-weight: 700;'>REDUCING AGENT</span>. <span style='color: #00d2ff; font-weight: 700;'>(1 mark)</span></div>
A4
[2 marks]
<div style='margin-bottom: 14px;'>A reducing agent is a substance that <span style='color: #ffffff; font-weight: 700;'>CAUSES</span> the reduction of another substance <span style='color: #00d2ff; font-weight: 700;'>(1 mark)</span>.</div><div style='margin-bottom: 14px;'>It does this by donating electrons / being oxidised itself.</div><div style='background: rgba(0, 210, 255, 0.08); padding: 18px; border-radius: 8px; border-left: 4px solid #00d2ff; line-height: 1.4;'><span style='color: #00d2ff; font-style: italic; font-weight: 600;'>Example:</span> Carbon acts as a reducing agent when it reduces iron oxide in the blast furnace:<br><span style='color: #ffffff; font-weight: 600; display: inline-block; margin-top: 8px;'>Fe₂O₃ + 3C → 2Fe + 3CO₂</span> <span style='color: #00d2ff; font-weight: 700;'>(1 mark)</span></div>
Part 3
Synthesis
Making Salts, Neutralisation & Salt Preparation Methods
What is a Salt?
Preparing Salts from Insoluble Bases (Core Practical)
Example: Making Copper Sulfate from Copper Oxide + Sulfuric Acid
Add EXCESS copper oxide (CuO) powder to warm dilute sulfuric acid (H₂SO₄) in a beaker. Stir continuously.
The solution turns BLUE as copper sulfate forms. Excess copper oxide remains — this shows all acid has been used.
FILTER the mixture through filter paper in a funnel to remove excess copper oxide.
Pour the blue copper sulfate filtrate into an evaporating dish.
Gently HEAT the solution to evaporate some water — stop when crystals begin to form at the edges.
Leave to COOL and CRYSTALLISE. Pat dry with filter paper.
CuO(s) + H₂SO₄(aq) → CuSO₄(aq) + H₂O(l)
⚠️ We use EXCESS base to ensure all the acid is neutralised.
Preparing Salts from Soluble Bases — Titration Method
Used when the base is SOLUBLE (e.g. sodium hydroxide, potassium hydroxide)
Fill a BURETTE with dilute acid. Fill a pipette with the alkali solution and transfer exact volume to a conical flask. Add 2-3 drops of INDICATOR.
Slowly add acid from burette to alkali, swirling constantly. Stop when the indicator changes colour (end point). Record the volume of acid used.
REPEAT the titration WITHOUT indicator using the exact same volumes. (Indicator would contaminate the salt.)
Transfer the neutralised solution to an evaporating dish. Heat gently to evaporate most of the water.
Leave to cool and crystallise. Collect and dry the pure salt crystals.
NaOH + HCl → NaCl + H₂O
(sodium hydroxide + hydrochloric acid → sodium chloride + water)
We repeat without indicator because we want a PURE salt — the indicator would contaminate it.
Solubility Rules
You need to know which salts are SOLUBLE and which are INSOLUBLE
Solubility rules help us choose the right METHOD for making a salt — insoluble bases use the excess method, soluble bases need titration.
📝 EXAM QUESTIONS — Synthesis (Set 1)
A student wants to make copper chloride salt. Name the acid and the base she should use, and write a word equation for the reaction.
[2 marks]
Describe how you would prepare pure, dry crystals of zinc sulfate starting from zinc oxide and dilute sulfuric acid. Your answer should include the reason why excess zinc oxide is added.
[4 marks]
A student adds calcium carbonate to hydrochloric acid. (a) Write a word equation for this reaction. [1 mark] (b) Describe two observations the student would make. [2 marks]
[3 marks]
Explain why a titration method must be used to prepare sodium chloride from sodium hydroxide and hydrochloric acid, rather than the excess base method.
[3 marks]
⏱ Suggested time: 12 minutes | ATTEMPT BEFORE TURNING OVER →
ANSWERS — Synthesis (Set 1)
[2 marks]
Hydrochloric acid (HCl)
Copper oxide (CuO)
Copper oxide + hydrochloric acid → copper chloride + water.
[4 marks]
Add excess zinc oxide to warm dilute sulfuric acid and stir.
Filter the mixture to remove excess zinc oxide.
Heat the filtrate (zinc sulfate solution) in an evaporating dish to concentrate it.
Allow to cool and crystallise; dry the crystals.
to ensure ALL the acid is neutralised / used up, so the final salt is not contaminated with acid. (already credited in mark scheme)
[3 marks]
Calcium carbonate + hydrochloric acid → calcium chloride + water + carbon dioxide.
Any two from:
Bubbles/fizzing observed (effervescence)
Solid calcium carbonate dissolves/disappears
The solution warms up slightly
CO₂ gas produced turns limewater milky (if tested)
[3 marks]
SOLUBLE base
so it cannot be filtered out if added in excess — it would contaminate the salt
Titration is used to find the exact volumes needed so that all the acid and alkali are neutralised completely, producing a pure salt.
Examiner Tip: In 4-mark method questions, aim for 4 distinct steps — each step = 1 mark!
- gcse-chemistry
- reactivity-series
- redox-reactions
- edexcel-chemistry
- science-revision
- salt-synthesis
- metal-extraction