Measuring Water Surface Tension: Capillary Rise Method
Learn to determine the surface tension of water using the capillary tube method with Jurin's Law, force derivations, and laboratory procedures.
Determination of Surface Tension of Water
Capillary Rise Method | Undergraduate Physics Laboratory
Theory: What is Surface Tension?
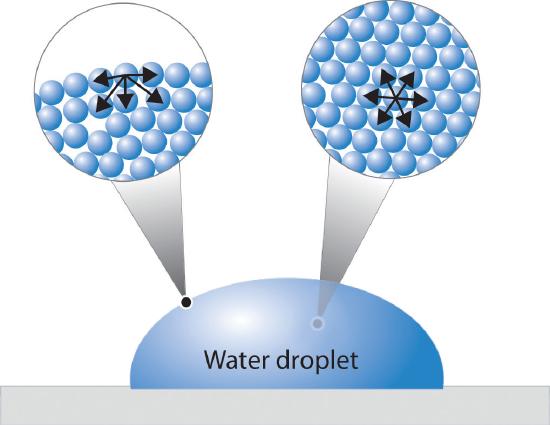
Surface tension (T) is a property of liquid surfaces that allows them to resist an external force. It arises due to the cohesive nature of liquid molecules.<br><br><b>Key Concepts:</b><br>• <b>Cohesion:</b> Attraction between like molecules (water-water).<br>• <b>Adhesion:</b> Attraction between unlike molecules (water-glass).<br>• In a capillary tube, adhesion > cohesion, causing the water to rise and form a concave meniscus.
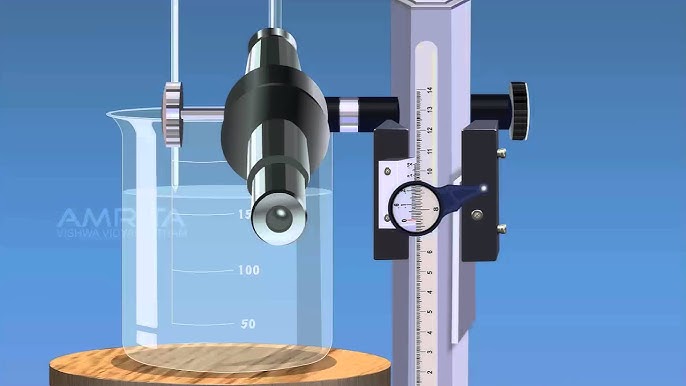
Apparatus Required
<ul><li><b>Capillary Tubes:</b> Fine uniform bore glass tubes.</li><li><b>Traveling Microscope:</b> For precise height measurement (0.001 cm precision).</li><li><b>Adjustable Stand:</b> To hold the capillary tube vertical.</li><li><b>Beaker & Pointer:</b> Clean glass beaker and an index pin.</li><li><b>Thermometer:</b> To record experimental temperature.</li><li><b>Cleaning Agents:</b> Chromic acid or Nitric acid, distilled water.</li></ul>
Theory: Jurin's Law
T = (r h ρ g) / (2 cosθ)
<div>Where:<br><b>T</b> = Surface Tension (N/m)<br><b>h</b> = Height of liquid rise (m)<br><b>r</b> = Radius of capillary bore (m)<br><b>ρ</b> = Density of liquid (kg/m<sup>3</sup>)<br><b>g</b> = Acceleration due to gravity (9.8 m/s<sup>2</sup>)<br><b>θ</b> = Angle of contact</div><br><div style='background:#e1f5fe; padding:15px; border-radius:5px;'><b>Assumption:</b> For a clean glass-water interface, the angle of contact θ is extremely small (≈ 0). Thus, <b>cosθ ≈ 1</b>.</div>
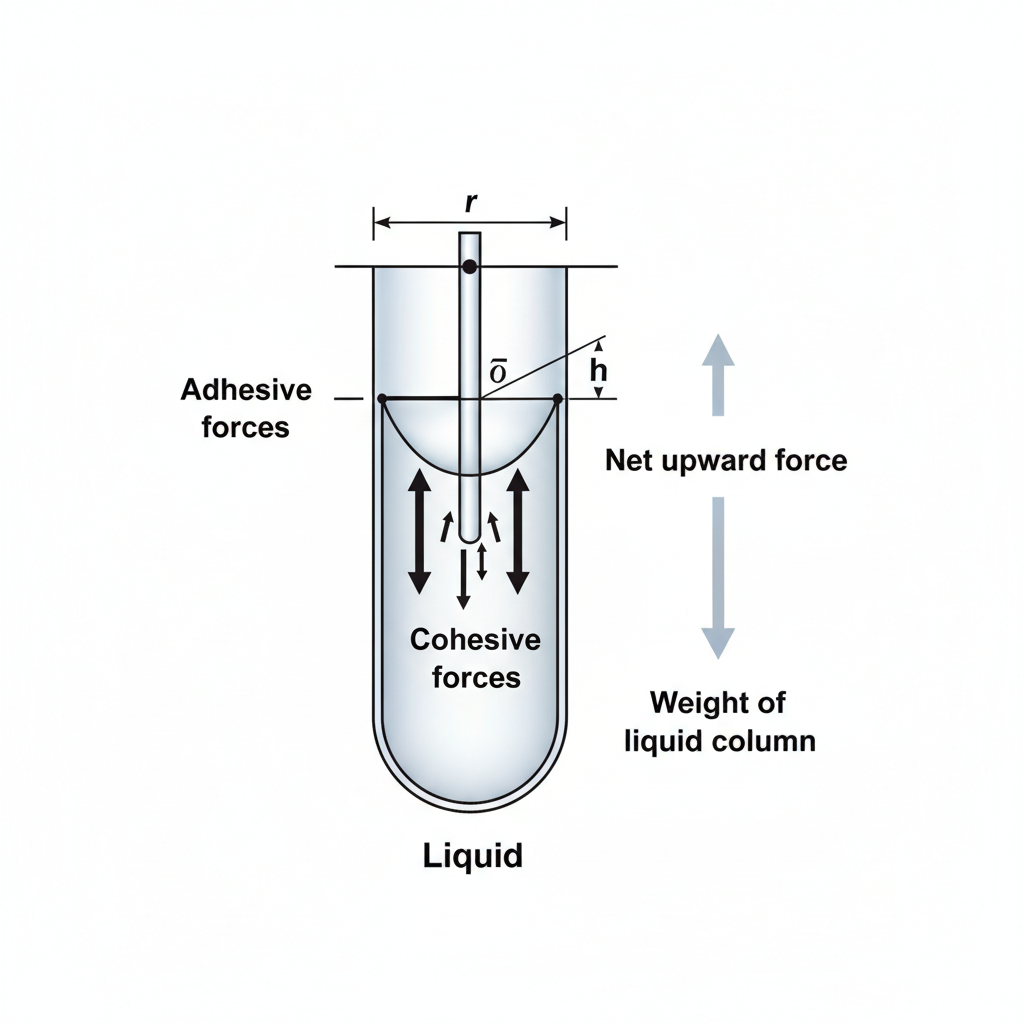
Force Balance Derivation
<b>1. Upward Force due to Surface Tension:</b><br>Surface tension T acts tangentially to the meniscus along the circumference (2πr). The vertical component upward is:<br><br><i>F<sub>up</sub> = (T cosθ) × (Circumference)</i><br><b style='color:#0277bd;'>F<sub>up</sub> = 2πr T cosθ</b>
<b>2. Downward Force (Weight of Column):</b><br>The weight of the liquid column of height h is mass × gravity. Mass = Volume × Density.<br><br><i>Volume = πr<sup>2</sup>h</i><br><b style='color:#c62828;'>W = πr<sup>2</sup>h ρ g</b>
<b>At Equilibrium: F<sub>up</sub> = W</b><br>2πr T cosθ = πr<sup>2</sup>h ρ g<br><br>Solving for T:<br><b>T = (r h ρ g) / 2 cosθ</b>
Diagram: Forces & Geometry
• <b>θ:</b> Contact angle<br>• <b>h:</b> Height of rise<br>• <b>T:</b> Surface Tension vector<br>• <b>mg:</b> Weight vector
Procedure: Setup & Cleaning
1. <b>Clean the Capillary:</b> Wash the capillary tube with chromic acid or nitric acid to remove grease, then rinse thoroughly with distilled water. <i>Caution: Grease alters the contact angle.</i>
2. <b>Clamp Vertically:</b> Clamp the capillary tube on the adjustable stand. Ensure it is perfectly vertical using a plumb line if available.
3. <b>Liquid Setup:</b> Fill a beaker with distilled water. Place it on the stand such that the capillary tube dips into the water. Adjust a pointed index pin (pointer) so its tip just touches the water surface outside the tube.
Procedure: Measurement
<ol><li><b>Focus Telescope:</b> Focus the traveling microscope on the meniscus of the water in the capillary tube. Align the horizontal cross-wire ensuring it is tangent to the meniscus curve. <b>Record Reading (A).</b></li><li><b>Focus on Pointer:</b> Lower the microscope to focus on the tip of the pointer touching the water surface. <b>Record Reading (B).</b></li><li><b>Calculate h:</b> Rise in capillary, <i>h = Reading A - Reading B</i>.</li><li><b>Find Radius (r):</b> Cut the tube at the meniscus level (or use method to focus on edges). Measure internal diameter in two perpendicular directions using the microscope cross-wires. Calculate mean radius.</li></ol>
Precautions & Sources of Error
<ul><li><b>Cleanliness is Crucial:</b> Presence of oil or grease drastically changes the surface tension.</li><li><b>Vertical Alignment:</b> The capillary tube must be perfectly vertical to ensure a uniform meniscus.</li><li><b>No Air Bubbles:</b> Ensure the liquid column contains no air bubbles.</li><li><b>Parallax Error:</b> Avoid parallax when reading the microscope scale.</li><li><b>Temperature Stability:</b> Surface tension decreases with temperature; minimize fluctuations during the experiment.</li></ul>
Observations & Calculation
<table style='width:100%; border-collapse:collapse; font-size:22px; text-align:center;'><thead><tr style='background:#0277bd; color:white;'><th style='padding:15px; border:1px solid #ccc;'>S.No</th><th style='padding:15px; border:1px solid #ccc;'>Microscope Reading<br>Meniscus (cm)</th><th style='padding:15px; border:1px solid #ccc;'>Microscope Reading<br>Pointer (cm)</th><th style='padding:15px; border:1px solid #ccc;'>Height<br>h = Difference (cm)</th><th style='padding:15px; border:1px solid #ccc;'>Radius<br>r (cm)</th><th style='padding:15px; border:1px solid #ccc;'>Surface Tension<br>T (dynes/cm or N/m)</th></tr></thead><tbody><tr><td style='padding:15px; border:1px solid #ccc;'>1</td><td style='padding:15px; border:1px solid #ccc;'>...</td><td style='padding:15px; border:1px solid #ccc;'>...</td><td style='padding:15px; border:1px solid #ccc;'>...</td><td style='padding:15px; border:1px solid #ccc;'>...</td><td style='padding:15px; border:1px solid #ccc;'>...</td></tr><tr><td style='padding:15px; border:1px solid #ccc;'>2</td><td style='padding:15px; border:1px solid #ccc;'>...</td><td style='padding:15px; border:1px solid #ccc;'>...</td><td style='padding:15px; border:1px solid #ccc;'>...</td><td style='padding:15px; border:1px solid #ccc;'>...</td><td style='padding:15px; border:1px solid #ccc;'>...</td></tr><tr><td style='padding:15px; border:1px solid #ccc;'>3</td><td style='padding:15px; border:1px solid #ccc;'>...</td><td style='padding:15px; border:1px solid #ccc;'>...</td><td style='padding:15px; border:1px solid #ccc;'>...</td><td style='padding:15px; border:1px solid #ccc;'>...</td><td style='padding:15px; border:1px solid #ccc;'>...</td></tr></tbody></table>
<b>Calculate Mean T:</b><br>1. Compute T for each trial using T = (rhρg)/2.<br>2. Take the average of calculated T values.<br>3. Ensure units are consistent (CGS: dynes/cm, SI: N/m).
Conclusion
The surface tension of water at room temperature was determined to be approximately <b>72.8 mN/m (at 20°C)</b>.<br><br>The experiment validates that measurement precision (especially of the hair-thin capillary bore radius) is critical for accurate results.
Height (h) is inversely proportional to radius (r).
- physics-experiment
- surface-tension
- capillary-rise
- fluid-mechanics
- undergraduate-physics
- jurin-law
- laboratory-guide