Targeting CTNNB1-S37F: TCR-T Cell Therapy for Solid Tumors
Discover how engineering T cells to target the public driver neoantigen CTNNB1-S37F offers a new precision immunotherapy approach for solid tumors.
Targeting CTNNB1-S37F: A Public Driver Neoantigen
Engineering T Cell Therapy for Solid Tumors
Background: Neoantigens & T Cell Therapy
Tumors carry somatic mutations which can produce neoepitopes.
Most mutations (< 3%) are rarely recognized by natural T cells.
TCR-engineered T cells can be designed to target specific neoantigens.
Strategy: Shift from 'Private' (patient-specific) to 'Public' (shared) antigens.
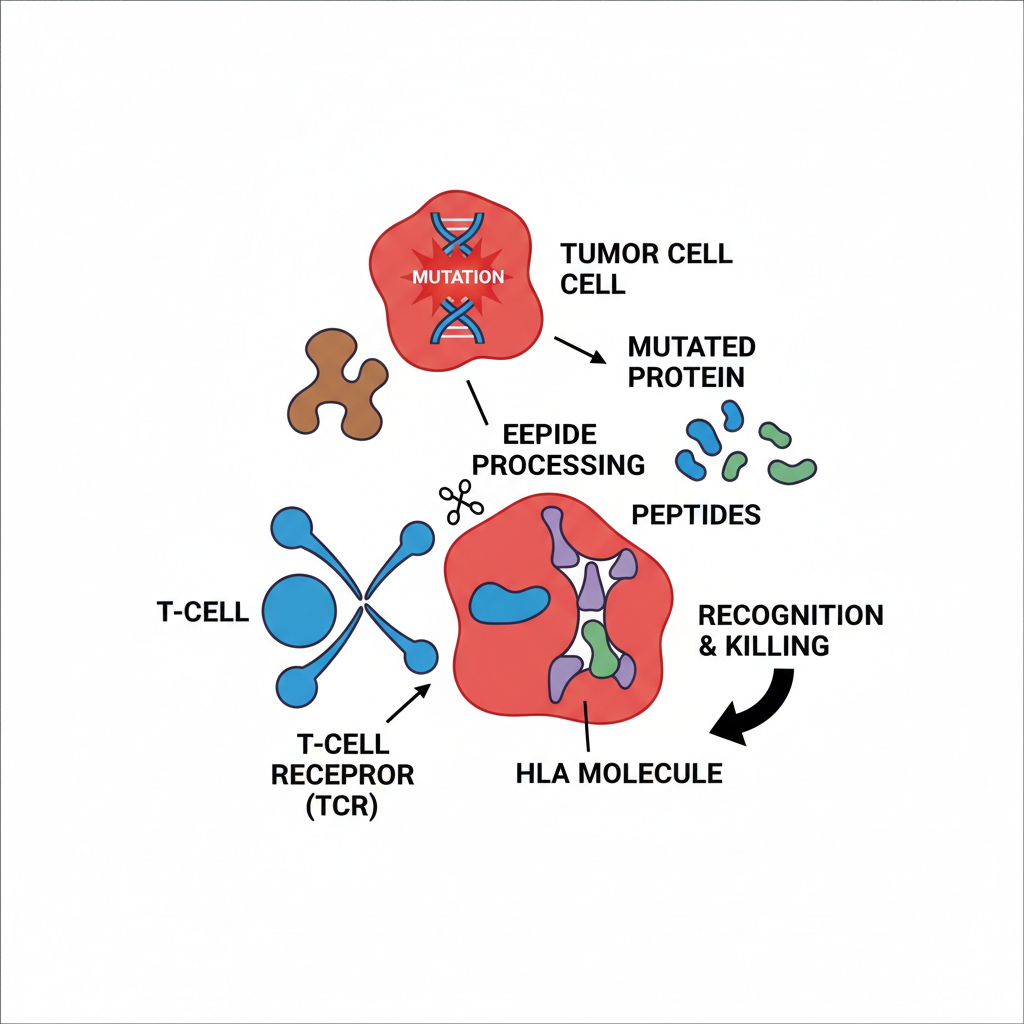
Neoantigens: How T cells see tumors
Driver mutations are essential for cancer growth and survival.
They are uniformly expressed, preventing immune escape/antigen loss.
Predecessors: KRAS and TP53 experimental therapies show promise.
Neoantigen = peptide created by tumor mutation
T cells cannot see proteins directly; they see peptides bound to HLA
HLA type determines presentation (not every patient presents the same mutation)
<strong>Private neoantigens:</strong> Unique to one patient
<strong>Shared neoantigens:</strong> Same mutation in many patients (e.g., CTNNB1-S37F)
Why CTNNB1 / β-Catenin?
CTNNB1 encodes β-catenin, a central component of the Wnt signaling pathway.
Mutated in ~3.2% of all cancers (hotspot in exon 3).
Mechanism: Mutation prevents degradation → Constitutive oncogenic signaling.
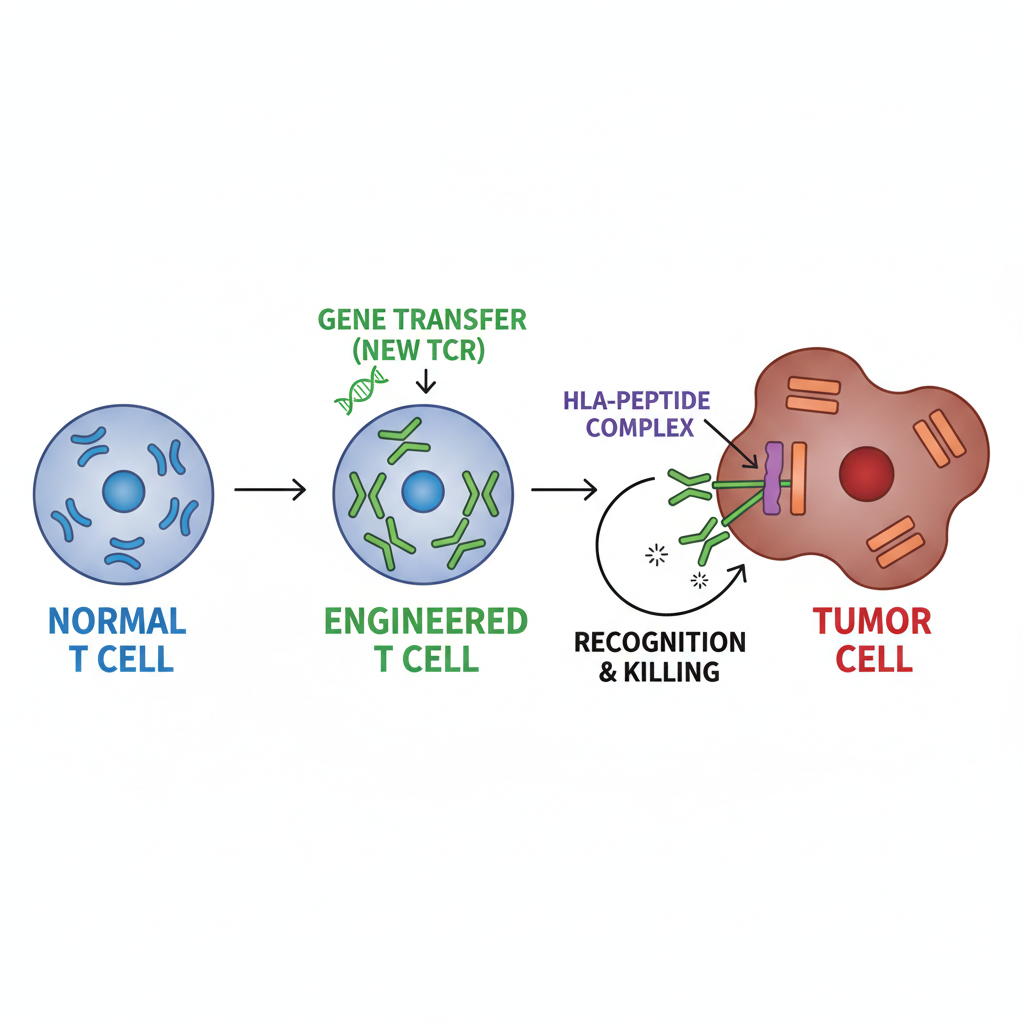
Engineering T cells to target CTNNB1-S37F
TCR-T cells: T cells engineered to express a specific TCR
Specific TCRs identified: <br>• TCRA2-2 for HLA-A*02:01 <br>• TCRA24 for HLA-A*24:02
Derived from healthy donors to avoid immune tolerance
Precision: Only T cells with the right HLA can recognize the peptide
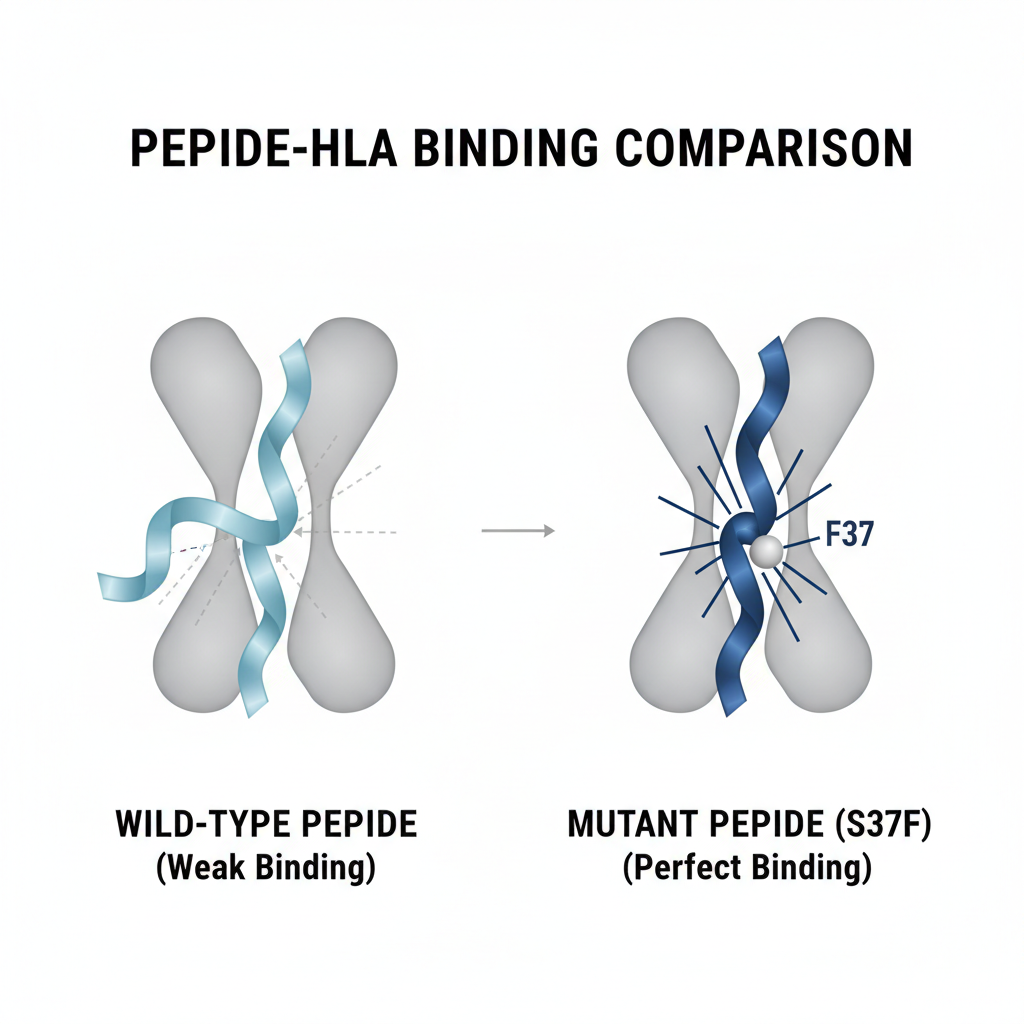
The Key Mutation: S37F
S37F is a recurrent hotspot mutation alongside S45F and T41A. It generates peptides predicted to bind with high affinity to common HLA alleles (HLA-A*02:01 and HLA-A*24:02).
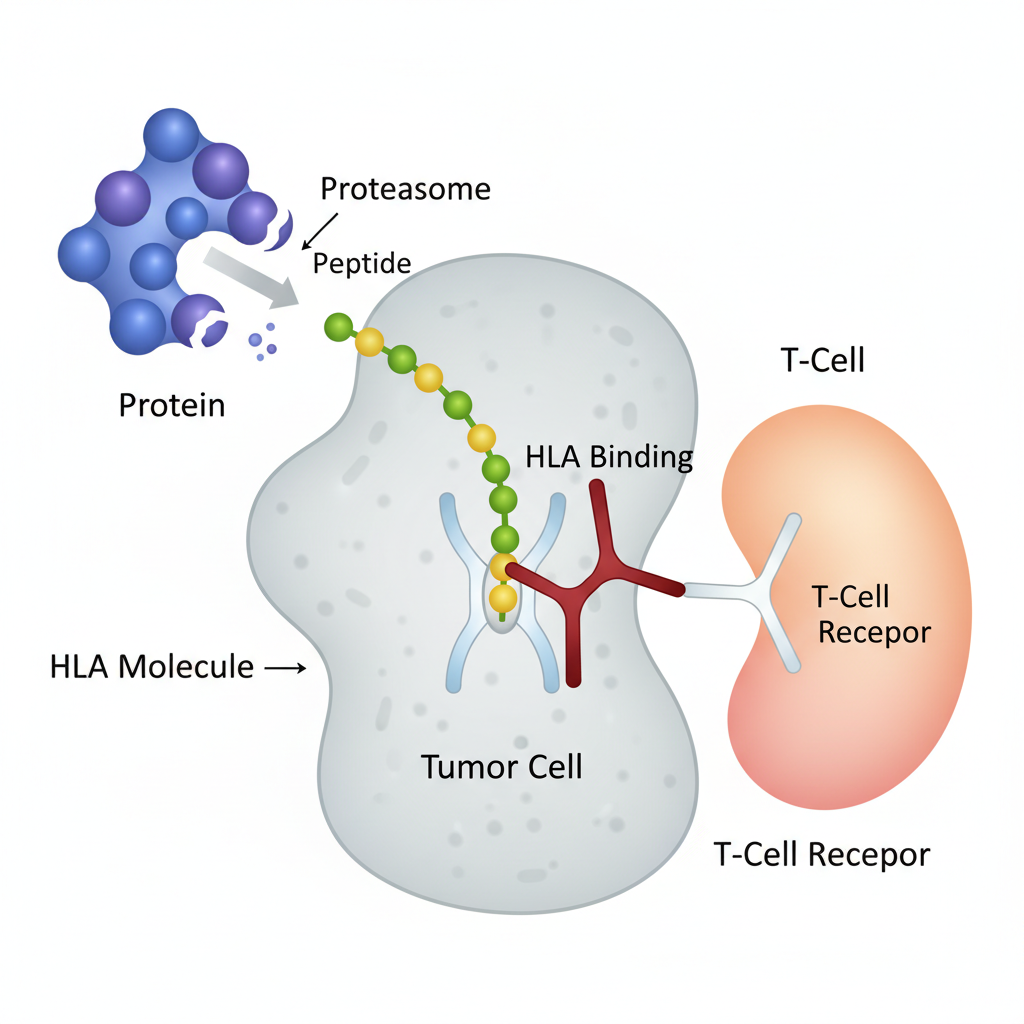
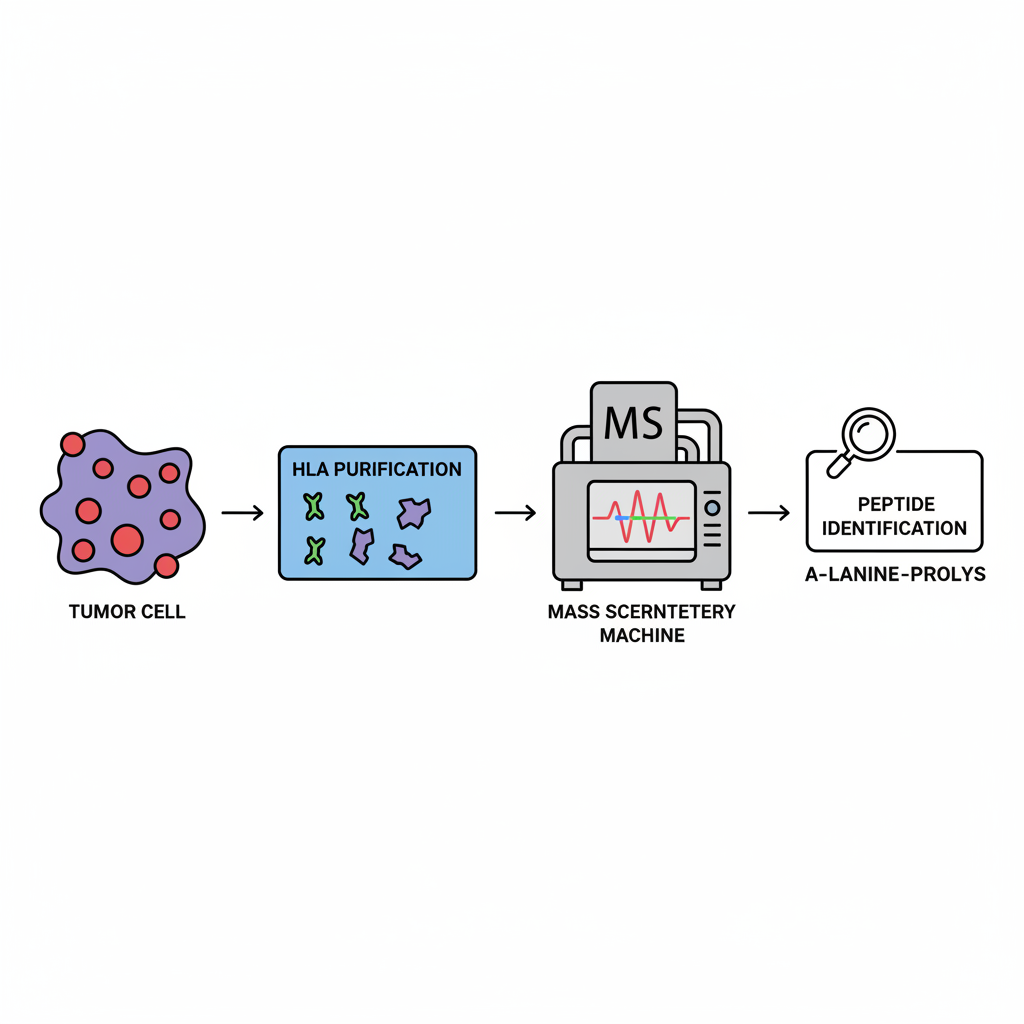
How do we know the peptide is actually presented?
Prediction is not enough: need to confirm peptide is physically on HLA
Mass spectrometry identifies physically presented peptides on HLA molecules
CTNNB1-S37F peptides were confirmed on tumor cells
Conclusion: They are visible targets for T cells
Confirming the Target Presentation
Mass spectrometry validated S37F peptide presentation on tumor surface.
HLA-A*24:02 : SYLDSGIHF
HLA-A*02:01 : YLDSGIHFGA
Central Question
Research Question
Can a shared oncogenic mutation generate naturally presented neoantigens that can be safely and effectively targeted by TCR-T cells?
Engineering S37F-Specific TCR-T Cells
Source: T cell receptors (TCRs) identified from healthy donors.
Engineering: Transducing T cells to express these high-affinity TCRs.
Mechanism: Specific recognition of tumor cells carrying the S37F mutation.
CTNNB1-S37F peptides are presented on HLA-A02:01 & A24:02
Mass spectrometry confirmed peptides are physically bound to HLA on tumor cells
<strong>Result:</strong> Only the mutant peptide was detected (WT peptide not recognized)
This confirms that T cells have a real, specific target to see
This differentiation is critical for safety (sparing healthy cells)
In Vivo Efficacy: Complete Eradication
Models: Melanoma (natural S37F) and Endometrial Adenocarcinoma (PDX). Results demonstrated rapid tumor regression compared to control groups.
Identifying TCRs that recognize CTNNB1-S37F
Naive T cells from HLA-matched healthy donors were primed with peptide
Reactive cells were sorted to identify specific TCR sequences
<strong>Found:</strong> TCRA2-2 (for HLA-A*02:01) and TCRA24 (for HLA-A*24:02)
High sensitivity: EC50 values indicate potent recognition
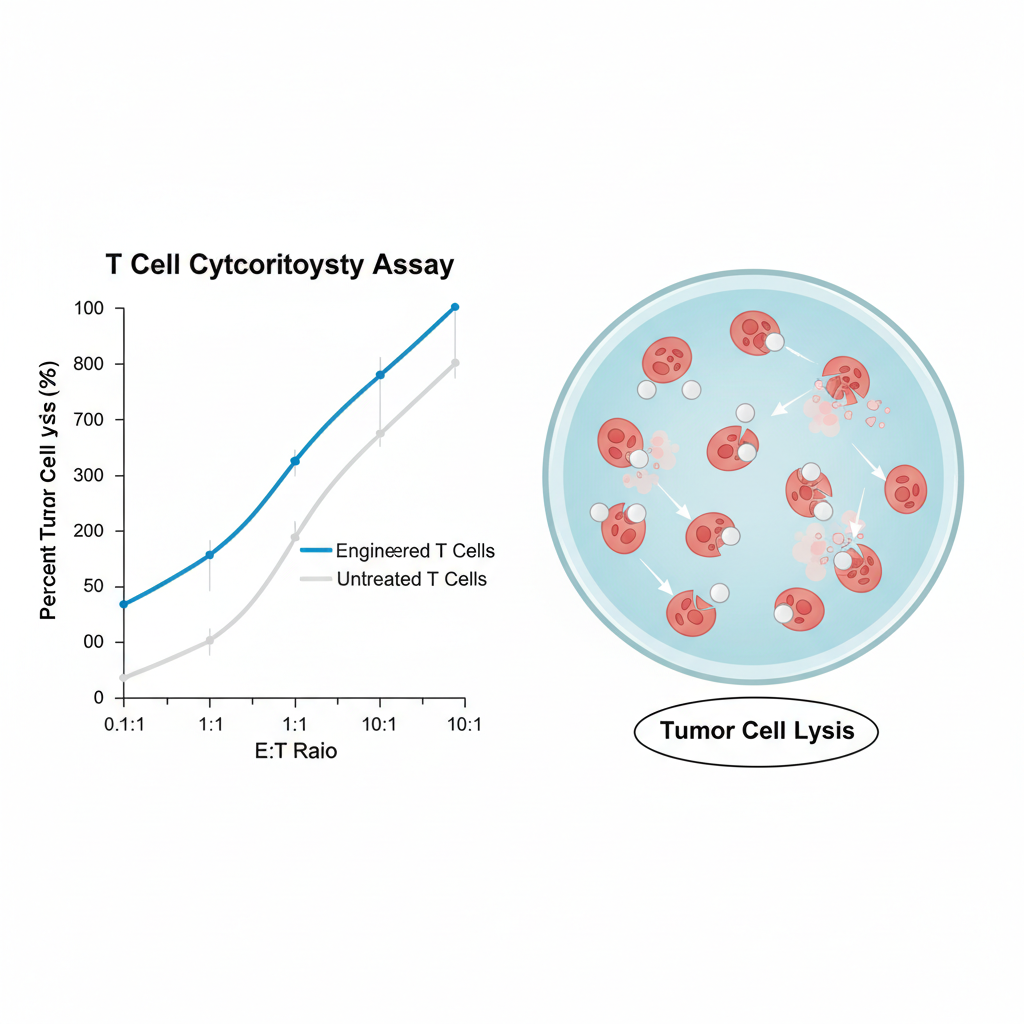
<strong>Action:</strong> In vitro killing of melanoma cell lines confirmed
Why This Matters: Addressing an Unmet Need
Target for 'Undruggable' Cancers: Providing options for β-catenin-mutated tumors resistant to standard drugs.
Recurrent & Public: A single therapy design could treat multiple patients effectively.
Proof of Concept: Validates TCR-T therapy for solid tumor driver mutations.
TCR-T cells eradicate tumors in mice & organoids
Mice with melanoma or PDX tumors treated with TCRA2-2 / TCRA24
<strong>Efficacy:</strong> 100% tumor-free early; long-term survival in 6/7 mice
Organoids (human tumors in vitro) also eradicated → confirms human relevance
<strong>No Exhaustion:</strong> Biomarkers (PD-1, TIM-3) remained low
Promising safety and durability profile
Summary & Take-Home Message
CTNNB1-S37F is a prime public driver neoantigen.
High-affinity TCRs can be isolated from healthy donors.
TCR-T cells achieve complete tumor eradication in vivo.
What we learned
Limitations & Considerations
<strong>HLA Restriction:</strong> Only a subset of patients (A*02:01 / A*24:02) can benefit
<strong>Models:</strong> Mice may not fully replicate human immune system complexity
PDX/organoids help but lack whole-body response dynamics
<strong>Heterogeneity:</strong> Combination therapy may be needed for long-term success
<strong>Clinical Translation:</strong> Patient selection is key (Mutation + HLA + Performance status)
Potential in Precision Immunotherapy
<strong>Scale:</strong> ~3,885 patients/year in US with CTNNB1-S37F + correct HLA could benefit
<strong>Strategy:</strong> Future therapies could combine multiple shared driver mutation TCRs
<strong>Vision:</strong> Paves the way for "Off-the-shelf" TCR-T libraries targeting solid tumors
- tcr-t-cell-therapy
- neoantigens
- ctnnb1-s37f
- cancer-immunotherapy
- precision-medicine
- solid-tumors
- oncology