Year 8 Science Revision: Physics, Biology & Chemistry
Comprehensive Year 8 Science revision guide covering Light and Space, Respiration, Unicellular Organisms, Combustion, Metals, and Geological Rock Cycles.
Year 8 Science Revision
Light & Space · Respiration · Unicellular Organisms · Combustion · Metals · Rocks
Year 8 · Science Department
What We'll Cover
Year 8 · Science Department
Light & Space
Earth & Space
Respiration & Breathing
Unicellular Organisms
Combustion & Metals
Rocks
01
Light on the Move
How light travels, what it is, and how it interacts with matter
Light on the Move
Key Terms
Vacuum
Completely empty space, no particles.
○
Matter
All things made of matter; 3 states: solid, liquid, gas.
◈
Transverse Wave
Vibrations at right angles to wave direction.
∽
Longitudinal Wave
Particles vibrate same direction as wave.
≣
Transparent
Light passes through without scattering.
◻
Translucent
Lets light through but scatters it.
▨
Opaque
Does not let light through.
◼
Reflect
Bounce off a surface.
↶
Absorb
Soak up / take in.
◉
Transmit
Pass through a substance.
➔
Year 8 Science
Rays, Images & Shadows
Ray
A narrow beam of light, represented by a straight line with an arrow showing its direction.
Image
A picture formed when light rays intersect or appear to intersect in a mirror, on a screen, or behind a lens.
Shadow
A dark area where light cannot reach because an opaque object is blocking the light path.
Pinhole Camera
A simple device without a lens that forms an inverted image when light travels through a tiny hole.
Source
The original point or object where a wave or light beam begins its journey.
Scattered
Light deflected from a straight path as it reflects off a rough surface or travels through an imperfect medium.
Reflection
Laws of reflection, ray diagrams, and types of reflection
02
Reflection — Key Terms & Law of Reflection
The Law of Reflection
Angle of Incidence = Angle of Reflection
Plane Mirror
Smooth flat mirror.
Ray Box
Equipment producing narrow light beam.
Normal
Imaginary line at right angles to surface where light hits.
Incident Ray
Ray going towards mirror.
Reflected Ray
Ray bouncing off mirror.
Angle of Incidence
Angle between incoming ray and normal.
Angle of Reflection
Angle between normal and leaving ray.
Ray Tracing
Method marking path of light ray.
SPECULAR REFLECTION
All reflected light goes same direction, smooth mirror.
DIFFUSE REFLECTION
Scattered in all directions, rough surface.
Year 8 Science
Refraction & Lenses
Refraction
Change in direction when light goes from one transparent material to another
Interface
Boundary between two materials
Lens
Curved transparent material that changes direction of light rays
Converging Lens
Lens that makes rays come together
Angle of Refraction
Angle between normal and refracted ray
Focal Point
Where parallel rays are brought together by converging lens
Focal Length
Distance between centre of lens and focal point
Cameras & the Human Eye
Year 8 Science
Digital Camera
uses electronics to record image
Sensor
detects light, converts to electrical signals
Retina
changes light energy into nerve impulses
Aperture
hole controlling how much light reaches sensor
Pupil
hole in front of eye that light passes through
Shutter
protects sensor, opens when photo is taken
Memory Card
stores images
03
Earth & Space
Astronomers, seasons, gravity, and the universe
Gathering the Evidence
History of Astronomy
Year 8 Science
Ptolemy
90–168 AD
Earth at the centre; Moon, Sun, and planets orbit around Earth.
Nicolaus Copernicus
1473–1543
Earth and planets orbit the Sun in circles.
Galileo Galilei
1564–1642
Used the telescope to observe the heavens, providing solid evidence for Copernicus' model.
Johannes Kepler
1571–1630
Sun at the centre; planets travel in <span style="color: #F4B41A; font-weight: 700;">elliptical</span> orbits, and moons orbit planets.
Seasons & Gravity
Year 8 Science
Beyond the Solar System
Year 8 Science
Constellation
Pattern of stars
Stars
Huge balls of gas giving out energy; Sun is a star
Galaxy
Large group of stars
Milky Way
Our galaxy containing our Sun
Universe
All millions of galaxies combined
Light Year
Distance light travels in 1 year ≈ 10 million million km
Proxima Centauri
Nearest star to Sun, 4.22 light years away
Phases of the Moon
Moon appears different shapes due to its position relative to Earth and Sun
Spacecraft
Allow scientists to investigate space by collecting samples and readings
04
Respiration
& Breathing
Aerobic & anaerobic respiration, gas exchange, and the circulatory system
Aerobic Respiration
Conducted the candle in a jar experiment.
Identified the active part of the air.
Proved oxygen is needed; showed it makes up 21% of air.
glucose <span style="color:#F4B41A">+</span> oxygen <span style="color:#F4B41A">→</span> carbon dioxide <span style="color:#F4B41A">+</span> water
Starting substances,<br>found on the left side of the equation.
New substances made,<br>found on the right side of the equation.
<strong style="color: #F4B41A; font-weight: 700;">Combustion vs. Respiration</strong> — same word equation, but an entirely different mechanism!
Using oxygen to release energy from glucose.
Same word equation as respiration but a different process.
Year 8 Science
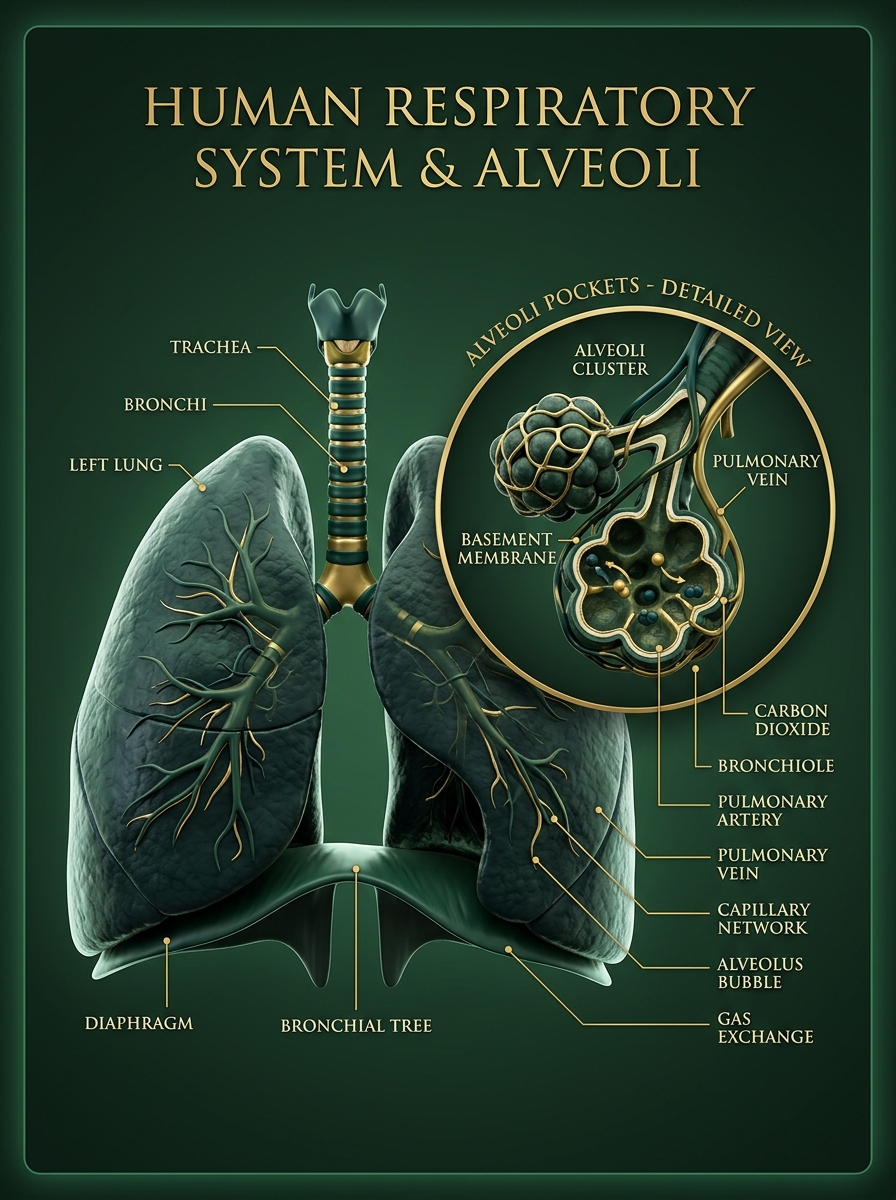
Gas Exchange System
Gas Exchange & Getting Oxygen
Year 8 Science
Breathing
Muscle movement allowing lungs to expand/contract
Ventilation
Movement of air in/out of lungs
Diaphragm
Organ below lungs, contracts/relaxes
Alveoli
Little pockets in lungs — increase surface area, walls one cell thick for fast diffusion
Diffusion
Movement of particles from high to low concentration
Mucus
Sticky liquid trapping dirt/microorganisms
Cilia
Tiny hairs sweeping mucus from lungs
Red Blood Cells
Haemoglobin
Oxygen binds here
Arteries
Capillaries
Veins
Plasma
Tissue Fluid
Anaerobic Respiration
Glucose
Lactic Acid
Anaerobic Process
Occurs in the cytoplasm when no oxygen is available, primarily during strenuous exercise.
Energy Yield
Anaerobic respiration releases significantly LESS energy compared to aerobic respiration.
Advantages
Provides a quick, sudden burst of energy exactly when the body forcefully demands it.
After Exercise
Lactic acid diffuses into the blood, is carried to the liver, and is converted back to glucose.
EPOC / Oxygen Debt
Extra oxygen needed after exercise to replace lost oxygen and convert lactic acid back to glucose.
Feature
Aerobic
Anaerobic
Oxygen
Required
Not required
Products
CO₂ + Water
Lactic Acid
Energy Yield
High
Low
Year 8 Science
05
Unicellular Organisms
Cells, microorganisms, fungi, bacteria, protoctists, and the carbon cycle
The Five Kingdoms of Life
UNICELLULAR
One cell (e.g. bacteria, amoeba)
MULTICELLULAR
Many cells (e.g. humans, plants)
PROKARYOTES
<div style='display: flex; flex-direction: column; gap: 12px; font-size: 19px;'><div style='display: flex; align-items: flex-start; gap: 12px;'><span style='color: #F4B41A; font-weight: bold;'>•</span> <span style='opacity: 0.9;'>Unicellular</span></div><div style='display: flex; align-items: flex-start; gap: 12px;'><span style='color: #F4B41A; font-weight: bold;'>•</span> <span style='opacity: 0.9;'>No nucleus</span></div></div>
PROTOCTISTS
<div style='display: flex; flex-direction: column; gap: 12px; font-size: 19px;'><div style='display: flex; align-items: flex-start; gap: 12px;'><span style='color: #AEE2D0; font-weight: bold;'>•</span> <span style='opacity: 0.9;'>Mainly unicellular</span></div><div style='display: flex; align-items: flex-start; gap: 12px;'><span style='color: #AEE2D0; font-weight: bold;'>•</span> <span style='opacity: 0.9;'>Have nucleus</span></div></div>
FUNGI
<div style='display: flex; flex-direction: column; gap: 12px; font-size: 19px;'><div style='display: flex; align-items: flex-start; gap: 12px;'><span style='color: #FF8C00; font-weight: bold;'>•</span> <span style='opacity: 0.9;'>Mainly multicellular</span></div><div style='display: flex; align-items: flex-start; gap: 12px;'><span style='color: #FF8C00; font-weight: bold;'>•</span> <span style='opacity: 0.9;'>Nucleus</span></div><div style='display: flex; align-items: flex-start; gap: 12px;'><span style='color: #FF8C00; font-weight: bold;'>•</span> <span style='opacity: 0.9;'>No own food</span></div></div>
PLANTS
<div style='display: flex; flex-direction: column; gap: 12px; font-size: 19px;'><div style='display: flex; align-items: flex-start; gap: 12px;'><span style='color: #32CD32; font-weight: bold;'>•</span> <span style='opacity: 0.9;'>Multicellular</span></div><div style='display: flex; align-items: flex-start; gap: 12px;'><span style='color: #32CD32; font-weight: bold;'>•</span> <span style='opacity: 0.9;'>Nucleus</span></div><div style='display: flex; align-items: flex-start; gap: 12px;'><span style='color: #32CD32; font-weight: bold;'>•</span> <span style='opacity: 0.9;'>Make own food</span></div></div>
ANIMALS
<div style='display: flex; flex-direction: column; gap: 12px; font-size: 19px;'><div style='display: flex; align-items: flex-start; gap: 12px;'><span style='color: #DC143C; font-weight: bold;'>•</span> <span style='opacity: 0.9;'>Multicellular</span></div><div style='display: flex; align-items: flex-start; gap: 12px;'><span style='color: #DC143C; font-weight: bold;'>•</span> <span style='opacity: 0.9;'>Nucleus</span></div><div style='display: flex; align-items: flex-start; gap: 12px;'><span style='color: #DC143C; font-weight: bold;'>•</span> <span style='opacity: 0.9;'>No own food</span></div><div style='display: flex; align-items: flex-start; gap: 12px;'><span style='color: #DC143C; font-weight: bold;'>•</span> <span style='opacity: 0.9;'>No cell wall</span></div></div>
Microorganisms
Too small to see without a microscope.
Diffusion
Particles spread out to fill the available space.
Viruses
Not living — cannot survive without a host cell.
Year 8 Science
Microorganisms in Detail
FUNGI
Budding
(asexual reproduction — small new cell grows from parent)
Fermentation
(anaerobic respiration: glucose → CO2 + water)
Population
Limiting Factor
BACTERIA
Binary Fission
(cell splits into two)
Chromosome
(instructions for organism)
Flagella
(tail for movement)
Lactic Acid
(glucose → lactic acid)
PROTOCTISTS
Algae
(uses photosynthesis)
Chloroplast
(site of photosynthesis)
Chlorophyll
(green substance absorbing light)
Producers
(make own food, start of food chain)
Year 8 Science
CO2 + water → glucose + oxygen
Decomposers & the Carbon Cycle
Photosynthesis
Respiration
Feeding
Decomposition
Combustion
Ecosystem
All environmental factors + organisms in a habitat
Decomposers
Feed on dead organisms/waste, recycle substances
Decay
Breakdown of dead organisms
Carbon Cycle
Shows how carbon compounds are recycled
Combustion
Burning fuels releasing CO₂
Feeding
Transfers carbon from plants to animals
Essential Nutrients
Carbohydrates
Energy source
Proteins
Growth and repair
Fats
Energy storage, insulator
Year 8 Science
Combustion
06
Fuels, burning, fire safety, air pollution, and global warming
Fuels & Oxidation
Year 8 Science
Fuel
Substance from which stored energy can be usefully transferred.
Fossil Fuels
Formed from living organisms millions of years ago — e.g., petrol, diesel.
Hydrocarbons
Compounds that contain only carbon and hydrogen atoms.
Combustion
Burning in air, gives out energy as heat and light.
Fuel Cell
Special cells that release energy directly from hydrogen.
Hydrogen
Oxygen
Water
Oxidation
The chemical process of reacting with oxygen.
Oxide
A chemical compound formed by the process of oxidation.
Reactants
The starting substances in a reaction.
Products
The new substances formed as a result.
Metal
Oxygen
Metal Oxide
Conservation of Mass
Mass is never gained or lost — atoms just rearrange.
Forms white zinc oxide. Mass appears to increase as oxygen from the air is added.
Fire Safety & Air Pollution
Year 8 Science
Global Warming & Climate Change
More CO₂ = Warmer Earth = Climate Change
Year 8 Science
Greenhouse Gases
Gases that trap energy from the Sun in the atmosphere (e.g., CO₂, Methane).
Greenhouse Effect
Energy trapped by greenhouse gases is transferred back to Earth's surface, causing warming.
Global Warming
The long-term increase in global temperatures due to a higher concentration of greenhouse gases.
Climate Change
Significant changes to global weather patterns, leading to more extreme storms, floods, and droughts.
Evidence
Average global temperatures are increasing, and polar ice caps are melting at a rapid rate.
07
Metals & Their Uses
Properties, corrosion, reactivity series, acids, and alloys
Year 8 · Science Department
Metal Properties & Corrosion
Physical Properties
Observable characteristics like melting point, density, and conductivity.
Chemical Properties
Describes how a substance acts and reacts with other substances.
Properties of Metals
Specific Metal Uses
Copper
Good conductor, used in circuits & water pipes. Unreactive, non-poisonous, malleable.
Aluminium
Strong and light, used in window frames.
Catalytic Converter
Platinum, palladium & rhodium — converts dangerous gases.
Corrosion
Any reaction with oxygen at the metal surface.
Rusting (Corrosion of Iron)
Requires both water AND oxygen.
iron + oxygen + water → iron hydroxide
Ti + O₂ → TiO₂
Preventing Rust (Barriers)
Year 8 Science
The Reactivity Series
Increasing Reactivity
Potassium
Sodium
Lithium
Calcium
Magnesium
Aluminium
Zinc
Iron
Tin
Lead
Copper
Mercury
Silver
Gold
Platinum
Reactivity
How quickly/vigorously something reacts.
Reactivity Series
List of metals in order of reactivity.
Metals & Water
Produce metal hydroxide + hydrogen;
sodium + water → sodium hydroxide + hydrogen
Effervescence
Production of gas when metals react with acid.
Reaction Key
Catches fire
Reacts very quickly
Reacts
Slow/partial
No reaction
Year 8 Science
Metals, Acids & Alloys
metal + acid → salt + hydrogen
magnesium + sulfuric acid → magnesium sulfate + hydrogen
Year 8 Science
Rocks
Rock types, weathering, erosion, the rock cycle, and materials in the Earth
08
Section 08 · Geological Sciences
Rocks & Their Properties
Geologist
Scientist studying rocks and Earth
Rocks
Naturally occurring substances made of different grains
Minerals
Chemical compounds found in rocks
Texture
Combination of sizes and shapes of grains
Interlocking Crystals
Grains fit together closely — firm, don't wear away easily
Rounded Grains
Gaps fall between grains — not strong, wear away easily
Porous
Rounded grain rocks that absorb and hold water
Permeable
Water is capable of running straight through
Uses of Rocks
Cement
Key building material made from limestone
Concrete
Composite mixture of cement + sand + gravel
Year 8 Science
Igneous & Metamorphic Rocks
Igneous Rocks
Metamorphic Rocks
Year 8 Science
Weathering & Erosion
CHEMICAL WEATHERING
Acidic rainwater reacts with minerals, wearing rocks away.
BIOLOGICAL WEATHERING
Plant roots splitting rocks apart.
PHYSICAL WEATHERING
Freeze-thaw action
(water in cracks → freezes → expands → crack gets bigger)
Temperature changes causing expansion and contraction.
Erosion
Movement of loose/weathered rock
Abrasion
Rock fragments bumping into each other
Sediment
Bits of rock/sand in streams
Glacier
Rivers of ice transporting large rocks
Year 8 Science
Sedimentary Rocks & The Rock Cycle
Sedimentary Rocks
Formed from layers of sediment experiencing compaction and cementation (e.g. sandstone, mudstone).
Compaction
Heavy overlaying layers create pressure that forces water out and squashes grains tightly together.
Cementation
Dissolved minerals crystallize in the gaps between sediments, acting as a natural glue.
Texture
Always composed of rounded grains that have been worn down by physical weathering and erosion.
Year 8 Science
Magma
Igneous Rock
Sediment
Sedimentary Rock
Metamorphic Rock
Materials in the Earth
Year 8 Science
Native State
Metals found as pure elements in rocks, requiring no chemical extraction.
Ores
Rocks containing enough metal or valuable compound worth mining commercially.
Extracting Ores
A multi-step process: Mining → Crushing → Chemical Reactions to purify.
Mining Problems
Destroys natural habitats on a massive scale and causes severe pollution.
Rare Metals
Excessively hard to obtain in high concentrations, making them very expensive.
Recycling
Using a material again rather than extracting raw ones or trashing them.
Recycling Advantages
Cuts down on mining pollution and landfill waste; allows raw material supplies to last longer; requires significantly less energy than extracting new ores.
Recycling: Better for the Planet!
Environment
Supply Lasts Longer
Less Energy
Revision Tips & Resources
Flash Cards
Write the term on one side, definition on the other
Word Banks
Create a glossary with all key definitions
Q Cards
Write questions and test yourself
Diagrams
Label diagrams: eye, lungs, rock cycle, etc.
BBC Bitesize • Seneca Learning
You've got this! Highlight each definition once you know it ✓
Year 8 · Science Department
- year-8-science
- science-revision
- ks3-science
- physics-revision
- biology-revision
- chemistry-revision
- space-and-light
- rock-cycle