Risks of Pathogen-Based Therapies: Safety & Regulation
Explore the clinical risks, viral shedding concerns, and regulatory hurdles of live biotherapeutics versus non-viral delivery alternatives.
The Case Against Pathogens as Therapies
Clinical Risks, Safety Challenges, and Regulatory Hurdles in Live Biotherapeutics
Executive Summary: Key Risk Domains
Direct Pathogenicity
Risk of reversion to virulence and uncontrolled replication in hosts.
Shedding & Transmission
Environmental contamination and secondary infection of non-patients.
Unpredictable Immunity
Cytokine storms, anaphylaxis, and neutralizing antibodies limiting efficacy.
Regulatory Complexity
High burden of proof for biosafety, containment, and long-term follow-up.
Primary Risk: Direct Pathogenicity
Even attenuated pathogens pose significant risks to patient safety. The mechanism of action often relies on replication, balanced delicately against safety.
Replication Competent Virus (RCV) risk due to recombination during manufacturing.
Genetic instability can lead to 'Reversion to Virulence' in vivo.
High variability in host response, especially in immunocompromised oncology patients.
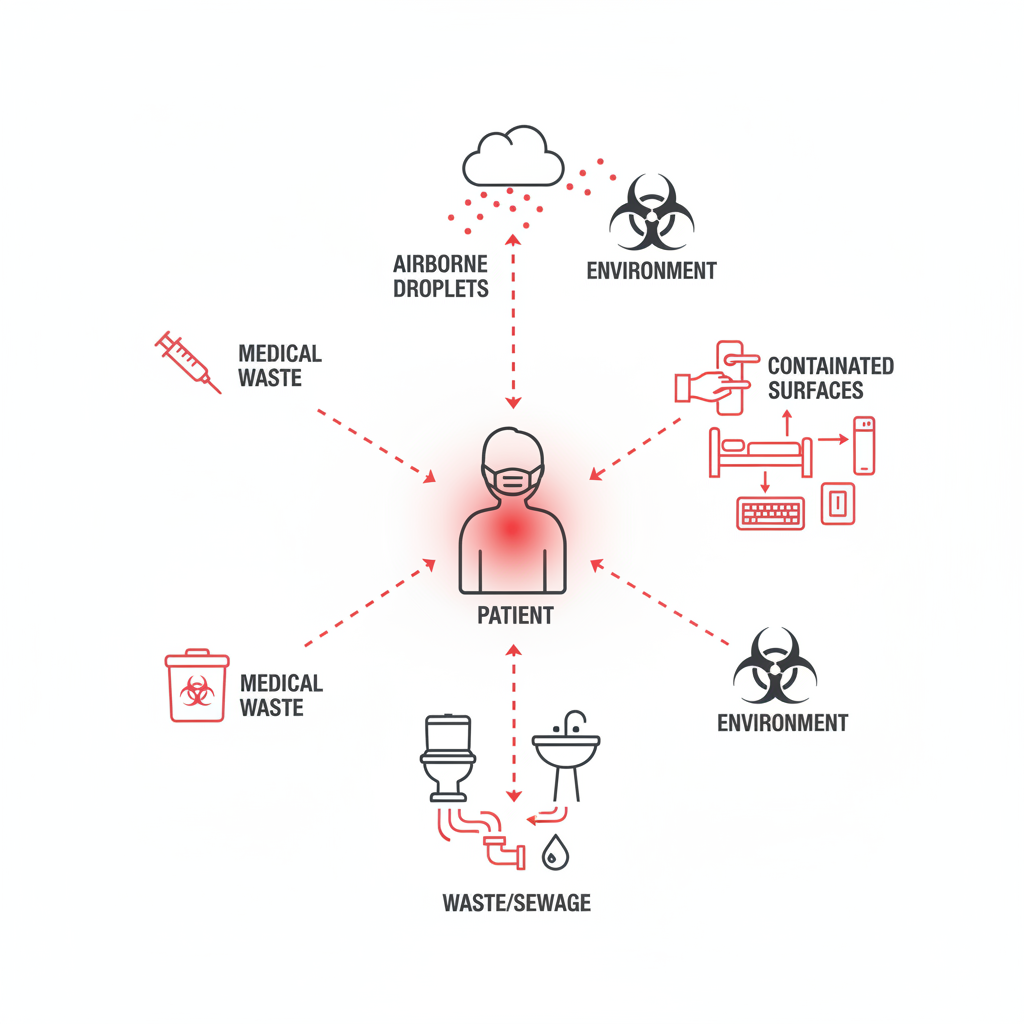
Shedding & Environmental Transmission
Unlike inert drugs, living therapies can escape the patient and infect others. Viral shedding remains a top regulatory concern.
Secondary Infection
Close contacts (family, healthcare workers) are at risk of inadvertent exposure.
Environmental Persistence
Viable virus detected in excreta/fluids for weeks post-administration requires strict isolation measures.
Representative Data: Viral Shedding Kinetics
Persistence of viral genomes in patient samples creates prolonged isolation requirements.
Unpredictable Immune Reactions
The immune system is designed to eliminate pathogens, creating a fundamental conflict with therapy.
Cytokine Release Syndrome (CRS): Potentially fatal systemic inflammation triggered by viral load.
Neutralizing Antibodies (NAbs): Pre-existing immunity can render the therapy useless before it acts.
Anaphylaxis: Immediate hypersensitivity reactions to viral capsid proteins.
Adverse Events Profile Comparison
Pathogen-based therapies demonstrate a distinct toxicity profile compared to non-viral modalities.
Regulatory & Safety Hurdles
Biosafety Requirements
Administration often requires BSL-2 or BSL-3 facilities, severely limiting the number of treatment centers and increasing costs.
Long-Term Follow-Up
FDA mandates up to 15 years of monitoring for gene therapy vectors to watch for delayed adverse events or insertional mutagenesis.
Manufacturing & CMC Challenges
Purity & Sterility: Difficult to sterilize live products without destroying efficacy.
Contamination Risk: Zero-tolerance for adventitious viruses or bacteria in bioreactors.
Stability: Complex cold-chain logistics required to maintain viral viability.
Safer Alternatives: Non-Viral Delivery
The industry is shifting towards synthetic vectors that offer efficacy without biological unpredictability.
Lipid Nanoparticles (LNPs): Validated by mRNA vaccines, low immunogenicity, scalable.
Antibody-Drug Conjugates (ADCs): Targeted delivery with established safety profiles.
Exosomes: Natural cellular transport without viral genetic baggage.
Conclusion: The Unfavorable Risk-Benefit Ratio
While conceptually appealing, pathogens as therapies introduce unmanageable variables into clinical practice. The dangers of infection, transmission, and immune toxicity outweigh the therapeutic potential, especially given the rise of safer synthetic alternatives.
- biotherapeutics
- gene-therapy
- viral-shedding
- biosafety
- clinical-risks
- biotechnology
- regulatory-affairs