In Vitro Skin Irritation Protocol: RhE Tissue Model Guide
Learn the 5-step SOP for In Vitro Skin Irritation testing using Reconstructed Human Epidermis (RhE). Covers MTT assay, GHS categories, and regulatory standards.
In Vitro Skin Irritation Protocol
Standard Operating Procedure for RhE Tissue Models
Protocol Overview
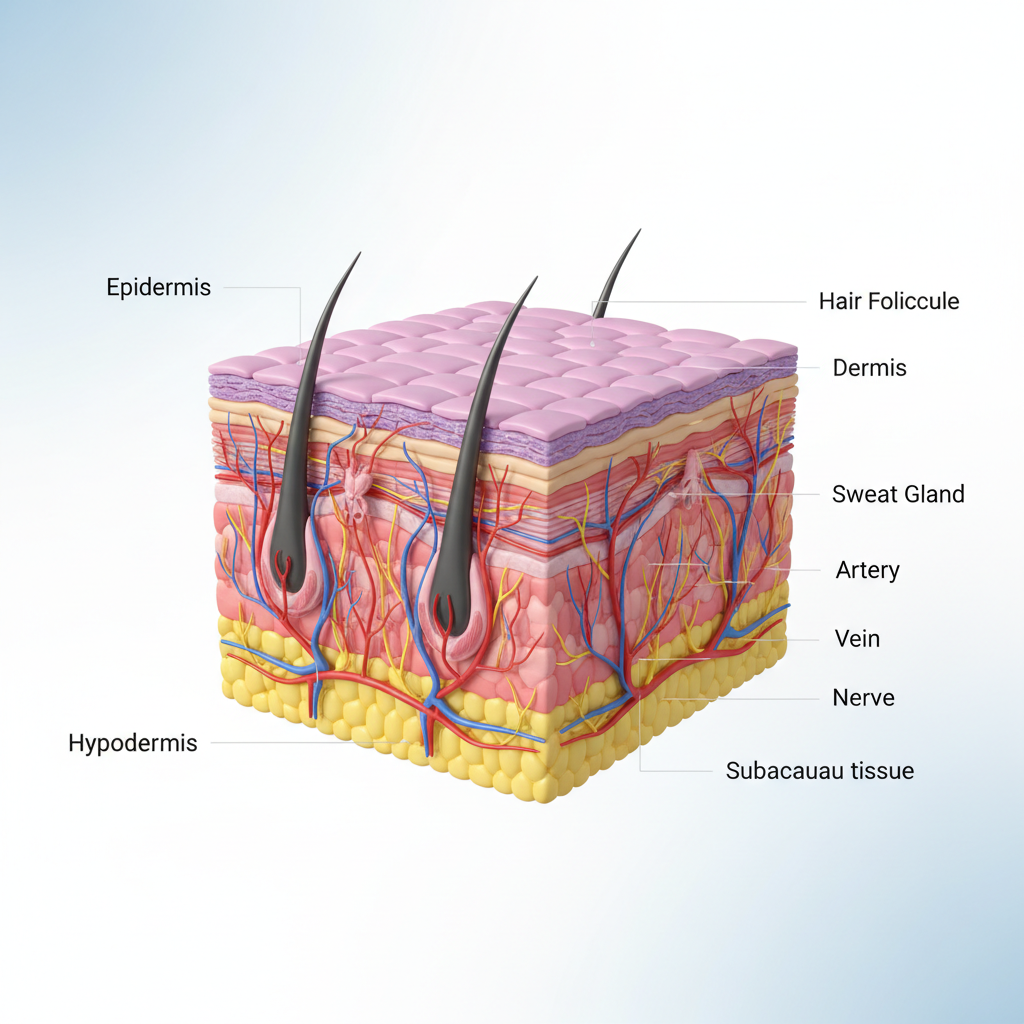
The In Vitro Irritation test generally utilizes Reconstructed Human Epidermis (RhE) designed to mimic the biochemical and physiological properties of human skin. This protocol quantifies skin irritation potential without animal testing.
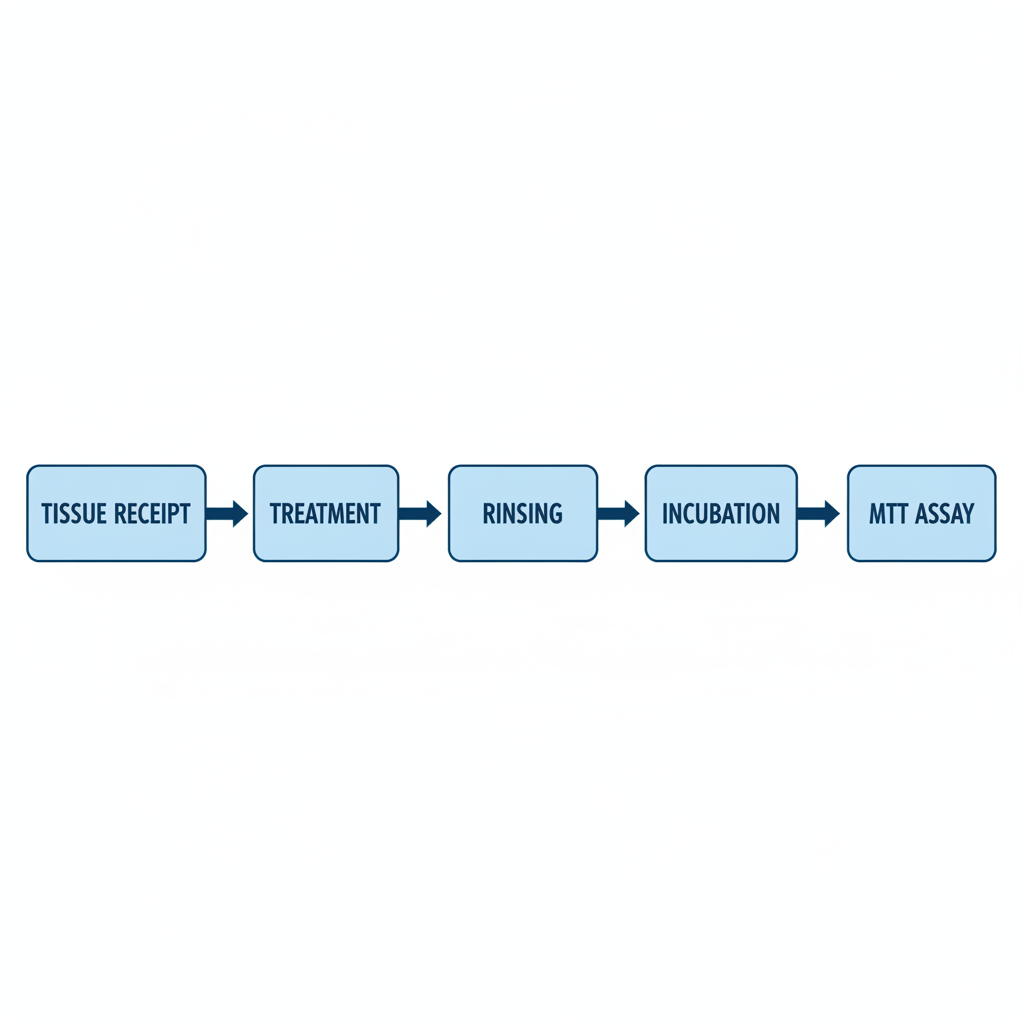
Step 1: Tissue Receipt & Conditioning
Initial incubation for 1 hour immediately upon receipt to equilibrate tissues.
Overnight incubation follows (with media change).
Environment: Standard culture conditions (37 ± 1°C, 5 ± 1% CO2 in humidified atmosphere).
Step 2: Tissue Treatment
Tissues are treated topically with the test substance to mimic real-world exposure.
Performed in triplicate tissues for statistical reliability.
Includes concurrent Negative Controls (NC) and Positive Controls (PC).
Step 3: Rinsing & Post-Expression
Tissue Rinsing
Remove test substance completely.
Initial Incubation
24 ± 1 hour post-treatment incubation.
Second Incubation
Fresh media change, then 42 ± 2 hours incubation to capture delayed effects.
Step 4: MTT Reduction Assay
Tissues are incubated with MTT solution after the post-treatment period.
Mechanism: Viable cells with active metabolism reduce MTT into a blue/purple formazan precipitate.
Non-viable cells do not convert the dye, remaining distinct.
Step 5: Extraction & Quantification
Isopropanol Extraction
Extract the purple formazan from tissues using isopropanol.
Spectrophotometry
Measure the Optical Density (OD) at 570 ± 30 nm.
Protocol Diagram Summary
From receipt to quantification, the entire process takes approximately 3-4 days including all incubation periods.
Prediction Model & Results
Non-Irritant (NI)
Mean tissue viability > 50%
Irritant (I) / GHS Cat 2
Mean tissue viability ≤ 50%
Regulatory Conclusion
This method aligns with UN GHS (Globally Harmonized System) standards.
Classifies substances into 'Category 2' (Irritant) or 'No Category' (Non-irritant).
Critical for safety datasheets (SDS) and occupational safety labeling.
- in-vitro-testing
- skin-irritation
- rhe-tissue-model
- mtt-assay
- laboratory-protocol
- un-ghs
- toxicology