Surface Tension of Water by Capillary Rise Method
Learn how to determine the surface tension of water using the capillary tube method, including theory, formulas, experimental procedure, and calculations.
Surface Tension of Water by Capillary Tube Method
Experiment No. 3
Physics Laboratory Presentation
Aim & Apparatus
<ul><li><strong>Aim:</strong> To determine the surface tension of water by capillary rise method.</li><li><strong>Apparatus:</strong></li><ul><li>Capillary tubes of uniform bore</li><li>A traveling microscope</li><li>Beaker with clean distilled water</li><li>Glass stand and clamp</li><li>Thermometer</li><li>reading lamp</li></ul></ul>
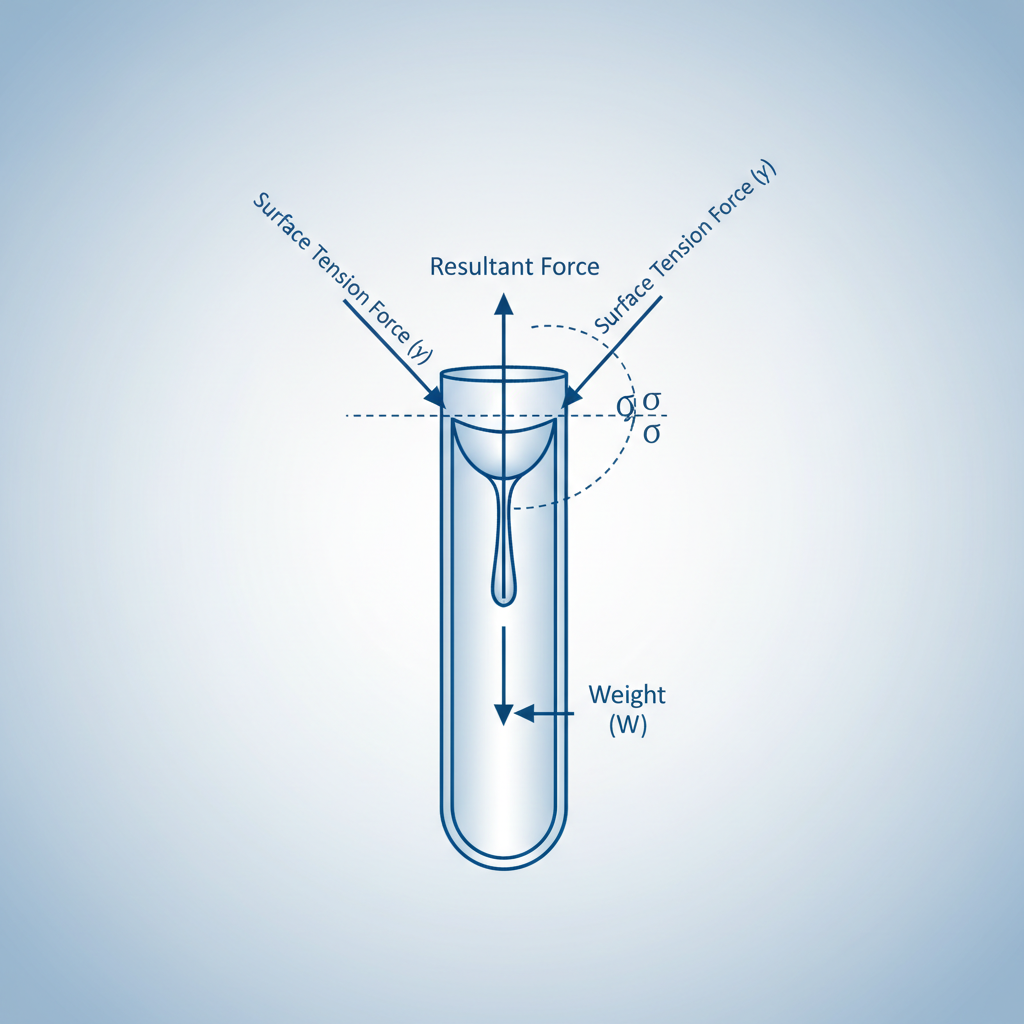
Theory: Force Analysis
The rise of liquid in the capillary is due to Surface Tension (T). At equilibrium, the upward force due to surface tension balances the downward weight of the liquid column.<br><br><strong>1. Upward Force ($F_{up}$):</strong><br>Acts along the tangent to the meniscus.<br>$F_{up} = (T imes 2 ho r) imes ext{vertical component}$<br>$F_{up} = 2 ho r T imes ext{cos} heta$
Theory: Derivation
<div style='background:#e0f2fe; padding:30px; border-radius:15px; border-left:8px solid #3b82f6;'><strong>2. Downward Weight ($W$):</strong><br>Weight of cylindrical column of height $h$.<br>$W = ext{Volume} imes ext{Density} imes g = ( ho r^2 h) imes ho imes g$<br><br><strong>Equilibrium Condition:</strong><br>$2 ho r T ext{cos} heta = ho r^2 h ho g$<br><br><strong>Final Formula:</strong><br>$T = rac{r h ho g}{2 ext{cos} heta}$</div>
Where:<br><strong>T</strong>: Surface Tension<br><strong>r</strong>: Radius of capillary<br><strong>h</strong>: Height of liquid rise<br><strong>ρ</strong>: Density of water ($10^3 kg/m^3$)<br><strong>θ</strong>: Angle of contact (approx 0° for water/glass)
Assumptions & Limitations
<strong>Assumptions:</strong><ul><li>The angle of contact $\theta$ is zero for clean water and glass.</li><li>The capillary tube has a uniform circular bore.</li><li>The temperature remains constant throughout the experiment.</li></ul>
<strong>Limitations/Factors:</strong><ul><li><strong>Temperature:</strong> Surface tension decreases as temperature increases.</li><li><strong>Impurities:</strong> Dissolved impurities (like grease or soap) drastically affect surface tension.</li><li><strong>Capillarity:</strong> If the tube is not perfectly vertical, $h$ readings will be erroneous.</li></ul>
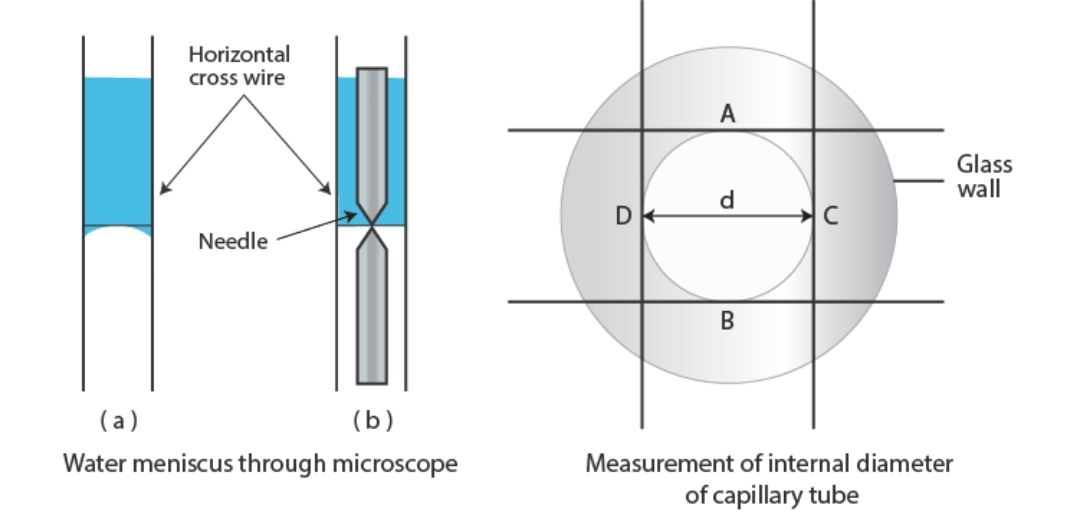
Experimental Diagram
Figure: (a) Setting cross-wire on the water meniscus. (b) Measurement of internal diameter of capillary tube using traveling microscope.
Procedure
1. Clean the capillary tube with acid and wash thoroughly with water.
2. Clamp the tube vertically and dip one end into a beaker of clean water. Allow water to rise.
3. Focus the traveling microscope on the meniscus. Read the main scale and vernier scale.
4. Remove the beaker and focus on the tip of a pointer adjusted to just touch the water surface to find height 'h'.
5. Cut the tube at the marked level (or simply use the microscope) to measure the internal diameter in two perpendicular directions.
Observations
<table style='width:100%; border-collapse:collapse; font-size:1.2rem; text-align:center;'><thead><tr style='background:#1e3a8a; color:white;'><th style='padding:15px;'>Measurement</th><th style='padding:15px;'>Reading (cm)</th><th style='padding:15px;'>Result</th></tr></thead><tbody><tr style='background:#e0f2fe;'><td style='padding:15px; text-align:left;'><strong>Meniscus Height (h)</strong></td><td></td><td></td></tr><tr><td style='text-align:left; padding:10px;'>Reading of Meniscus</td><td>12.45</td><td rowspan='2' style='vertical-align:middle; font-weight:bold;'>h = 3.20 cm</td></tr><tr><td style='text-align:left; padding:10px;'>Reading of Pointer</td><td>9.25</td></tr><tr style='background:#e0f2fe;'><td style='padding:15px; text-align:left;'><strong>Diameter (d)</strong></td><td></td><td></td></tr><tr><td style='text-align:left; padding:10px;'>Horizontal Diameter ($d_1$)</td><td>0.098</td><td rowspan='3' style='vertical-align:middle; font-weight:bold;'>r = 0.049 cm</td></tr><tr><td style='text-align:left; padding:10px;'>Vertical Diameter ($d_2$)</td><td>0.098</td></tr><tr><td style='text-align:left; padding:10px;'>Mean Diameter</td><td>0.098</td></tr></tbody></table>
<strong>Constants:</strong><br>Temperature: 20°C<br>Density of water ($ ho$): $1 g/cm^3$ or $1000 kg/m^3$<br>Acceleration due to gravity ($g$): $980 cm/s^2$ or $9.8 m/s^2$
Calculation
$T = \frac{r h \rho g}{2}$
Using the sample data:<br><ul><li>Radius ($r$) = $0.049 \text{ cm}$ = $0.00049 \text{ m}$</li><li>Height ($h$) = $3.20 \text{ cm}$ = $0.032 \text{ m}$</li><li>Density ($\rho$) = $1000 \text{ kg/m}^3$</li><li>Gravity ($g$) = $9.8 \text{ m/s}^2$</li></ul><br>Substitution:<br>$T = \frac{(0.00049)(0.032)(1000)(9.8)}{2}$<br>$T = \frac{0.153664}{2} = 0.0768 \text{ N/m}$
(Calculation in CGS Units yields dynes/cm, then convert to SI)
Error Analysis & Result
<strong>Percentage Error:</strong><br>Standard Value of T at 20°C $\approx 0.0728$ N/m.<br>Experimental Value $T_{exp} = 0.0768$ N/m.<br>$$\text{Error} = \frac{|0.0768 - 0.0728|}{0.0728} \times 100 \approx 5.5\%$$<br><br><strong>Sources of Error:</strong><br>1. Non-uniformity of the capillary bore.<br>2. Error in measuring meniscus height (parallax).<br>3. Presence of microscopic impurities affecting contact angle.
<strong>FINAL RESULT</strong><br>The surface tension of water was determined to be:<br><span style='font-size:2.5rem; color:#1e3a8a;'>0.0768 N/m</span>
References
<ul><li>Resnick, Halliday, Walker. <em>Fundamentals of Physics</em>. Wiley.</li><li>NCERT Physics Laboratory Manual.</li><li>Measurement Technique Instructions, 2026.</li></ul>
- physics-experiment
- surface-tension
- capillary-rise
- fluid-mechanics
- laboratory-manual
- science-presentation