Nematodes: Biology, Impact & Meloidogyne incognita Overview
Learn about the biology, classification, and crop impact of Root-Knot Nematodes. Detailed guide on life cycles, infection mechanisms, and management.
NEMATODES
Biology, Classification, Crop Impact & Meloidogyne incognita
A Comprehensive Scientific Overview
Plant Pathology | Nematology | Crop Science
CONTENTS
01
Introduction to Nematodes
02
Scientific Classification
03
Types of Plant-Parasitic Nematodes
04
Symptoms in Crops
05
Underground Symptoms
06
Life Cycle Overview
07
Life Cycle – Stage by Stage
08
Lab Assay Methods
09
Baermann Funnel Technique
10
Root Staining & Microscopy
11
<i>Meloidogyne incognita</i> – Introduction
12
<i>M. incognita</i> – Infection Mechanism
13
Giant Cell Formation
14
Economic Impact
15
Management & Control
INTRODUCTION TO NEMATODES
What are Nematodes?
Microscopic roundworms belonging to phylum Nematoda; among the most abundant multicellular animals on Earth with over 25,000 described species.
Size & Structure
Typically 0.3–3 mm long; unsegmented, bilaterally symmetrical, thread-like body with a complete digestive system.
Habitat
Found in nearly every ecosystem: soil, freshwater, marine, and as parasites of plants and animals.
Plant-Parasitic Nematodes
~4,100 species parasitize plants; cause $80–173 billion in global crop losses annually.
Mode of Feeding
Use a hollow needle-like stylet to pierce plant cells and extract nutrients.
Plant Pathology | Nematology | Crop Science
SCIENTIFIC CLASSIFICATION
Taxonomic Hierarchy of Nematodes
Kingdom
Animalia
Phylum
Nematoda
Class
Chromadorea
Order
Rhabditida
Family
Meloidogynidae
Genus
Meloidogyne
Species
M. incognita
Key Facts
Phylum
Nematoda
contains over 25,000 described species.
Plant-parasitic nematodes belong to Class
Chromadorea
.
TYPES OF PLANT-PARASITIC NEMATODES
Over 4,100 species of plant-parasitic nematodes identified globally
ROOT-KNOT NEMATODES
(Meloidogyne spp.)
Sedentary endoparasites
Form characteristic galls on roots
Most economically important group worldwide
CYST NEMATODES
(Heterodera & Globodera spp.)
Female body hardens into protective cyst after death
Contains hundreds of eggs
Affects cereals and potatoes
LESION NEMATODES
(Pratylenchus spp.)
Migratory endoparasites
Move through root tissue causing dark necrotic lesions
Wide host range
STEM & BULB NEMATODES
(Ditylenchus spp.)
Attack stems, bulbs and leaves
Cause stunting and distortion
Important in bulb crops like onion and garlic
SYMPTOMS IN CROPS
Above-Ground Indicators
STUNTED GROWTH
Plants appear shorter than normal; reduced internode elongation due to impaired nutrient uptake from damaged roots.
WILTING
Plants wilt during daytime even with adequate soil moisture; root damage reduces water absorption capacity.
YELLOWING (Chlorosis)
Leaves turn yellow resembling nitrogen/iron deficiency; caused by reduced nutrient translocation from infested roots.
REDUCED YIELD
Up to 80% yield loss in tomatoes; 41–88% in cassava storage roots under heavy infestation.
MIMICS DEFICIENCY
Symptoms often confused with nutrient deficiency or drought stress, delaying diagnosis.
Annual global crop losses: $80–173 billion USD
ROOT SYMPTOMS & UNDERGROUND DAMAGE
GALL FORMATION
Characteristic bead-like swellings (galls/knots) on roots caused by giant cell formation; hallmark of Meloidogyne infection.
ROOT NECROSIS
Dark brown lesions and rotting of root tissue; caused by migratory nematodes like Pratylenchus.
FORKED/DISTORTED ROOTS
Root crops like carrots become forked, stubby, or hairy; reduces marketability.
REDUCED ROOT MASS
Overall root biomass decreases; fewer lateral roots; compromised water/nutrient uptake.
SECONDARY INFECTIONS
Damaged roots become entry points for fungi and bacteria, compounding damage.
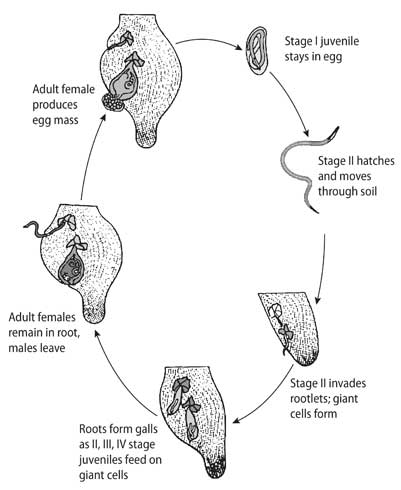
LIFE CYCLE OF MELOIDOGYNE INCOGNITA
Completes in 21–30 days at 25–28°C
EGG STAGE
Female lays 300–500 eggs in gelatinous matrix outside root
J1 JUVENILE
First molt occurs inside egg; vermiform stage
J2 JUVENILE
Hatches, migrates through soil to find root
J3 & J4
Sedentary molting stages inside root gall
ADULT FEMALE
Sedentary, pear-shaped, remains in root, lays eggs
ADULT MALE
Vermiform, leaves root, does not feed
Optimal temperature: 28°C | Cycle duration: 3 weeks | Each female: up to 1,500 eggs
LIFE CYCLE STAGES – DETAILED BREAKDOWN
EGG
Laid in gelatinous matrix;<br>embryogenesis occurs inside
J1
First-stage juvenile;<br>first molt occurs within egg shell
J2<br><span style='font-size: 18px; color: #fdf2e9; opacity: 0.9;'>(infective)</span>
Hatches; moves through soil;<br>uses stylet to invade root tips;<br><b>only free-living infective stage</b>
J3 & J4
Sedentary inside developing gall;<br>molts twice;<br>feeding on giant cells
ADULT<br><span style='font-size: 20px;'>FEMALE</span>
Sedentary; swells to pear shape;<br>produces 300–1,500 eggs in<br>gelatinous matrix on root surface
ADULT<br><span style='font-size: 20px;'>MALE</span>
Reverts to vermiform; exits root;<br>fertilizes female (optional);<br>dies shortly after
<span style='color: #f1c40f; font-weight: 800;'>J2</span> is the ONLY soil-mobile infective stage <span style='color: #a3e4d7; margin: 0 15px;'>|</span> <span style='color: rgba(255,255,255,0.9);'>Stylet length: ~14–16 µm</span>
LABORATORY ASSAY METHODS
Extracting Nematodes from Plant Roots & Soil
BAERMANN FUNNEL
Cut roots 2–3 cm, place on tissue mesh over funnel with water, extract after 24–72 hrs. Best for active vermiform J2 stage. Passive migration method.
BLENDER MACERATION
Blend roots in NaOCl solution 3 min, sieve through 170 and 500 mesh. Recovers all life stages including eggs. Highest total recovery method.
MIST CHAMBER / SHAKER
Washed roots in solution, incubated 72 hrs with gentle agitation, sieve collected. Best for delicate nematode specimens.
Standardize sample: 50 cc root volume per extraction
Extraction Method
Recovery Efficiency Comparison
Blender Maceration
Highest
Mist Chamber
Medium
Baermann Funnel
Lowest (Total)
ROOT STAINING & MICROSCOPY
Visualizing Nematodes in Root Tissue
ACID FUCHSIN STAINING (Byrd et al. 1983)
Most common root-staining protocol
Roots boiled in lactophenol-acid fuchsin; destained in glycerol
Nematodes stain bright red/pink against clear root tissue
Used for: counting, identifying life stages in situ
LACTOPHENOL COTTON BLUE
Stains nematode body blue for microscopy
Used for: slide mounts, detailed morphology study
LIGHT MICROSCOPY
40x–400x magnification for identification
Measure stylet length, body dimensions, tail shape for species ID
COBB'S DECANTING & SIEVING
Soil extraction; 250–600 cc soil subsamples
Pour through 60-mesh sieve 5 times between pitchers
Collect and count nematodes per 100g soil
Proper fixation: 4% formaldehyde or TAF (triethanolamine-formalin-acetic acid) for permanent mounts
MELOIDOGYNE INCOGNITA
Southern Root-Knot Nematode
COMMON NAME
Southern Root-Knot Nematode
CLASSIFICATION
Kingdom Animalia → Phylum Nematoda → Class Chromadorea → Genus Meloidogyne → Species <i>M. incognita</i>
DISTRIBUTION
Worldwide; most damaging in tropical & subtropical regions; soil temperatures 25–32°C optimal
HOST RANGE
Extremely wide — over 2,000 plant species including tomato, tobacco, cotton, soybean, cucumber, pepper
REPRODUCTION
Primarily parthenogenetic (no male needed); mitotic parthenogenesis; produces clonal offspring
DISCOVERY
First described by Kofoid & White (1919)
One of the most economically significant plant pathogens globally
INFECTION MECHANISM OF M. INCOGNITA
How Root-Knot Nematodes Invade and Hijack Plant Cells
HATCHING & SOIL MIGRATION
J2 juvenile hatches from egg in soil. Detects root exudates (CO2 gradients). Moves through soil toward root tips. Distance: up to 60 cm.
ROOT PENETRATION
J2 uses hollow stylet (14–16 µm) to mechanically pierce root cap cells near the elongation zone. Injects esophageal gland secretions into cells.
MIGRATION IN ROOT
Moves intercellularly through cortex. Heads toward vascular cylinder. Becomes sedentary once feeding site is established.
GIANT CELL INDUCTION
Effector proteins reprogram 5–7 vascular cells to fuse into multinucleate giant cells (feeding sites). Cells enlarge 100x and repeatedly divide nuclei without wall formation.
GALL FORMATION
Surrounding cells undergo hypertrophy, visible gall forms. Female swells to pear shape, lays 300–1,500 eggs in gelatinous matrix. Cycle repeats.
KEY EFFECTORS
16D10, 19C07, MiPFN — reprogram plant cell cycle and gene expression
GIANT CELL FORMATION
The Nematode's Permanent Feeding Strategy
🔬 WHAT IS A GIANT CELL?
Multinucleate, hypertrophied plant cell derived from vascular parenchyma. Contains 8–100+ nuclei. Serves as the nematode's permanent nutrient sink.
⚙️ HOW ARE THEY FORMED?
Nematode injects effector proteins via stylet. Effectors suppress plant immunity (salicylic acid pathway). Trigger repeated nuclear divisions without cytokinesis (karyokinesis without cell plate). Cell walls dissolve partially; cells fuse.
🌿 PLANT CELL CHANGES:
<div style="margin-bottom: 6px; padding-left: 20px; text-indent: -20px;"><strong style="color:#2d6a4f; margin-right: 6px;">—</strong>Cell volume increases up to 100-fold</div><div style="margin-bottom: 6px; padding-left: 20px; text-indent: -20px;"><strong style="color:#2d6a4f; margin-right: 6px;">—</strong>Dense cytoplasm with many organelles</div><div style="margin-bottom: 6px; padding-left: 20px; text-indent: -20px;"><strong style="color:#2d6a4f; margin-right: 6px;">—</strong>Increased metabolic activity</div><div style="padding-left: 20px; text-indent: -20px;"><strong style="color:#2d6a4f; margin-right: 6px;">—</strong>Acts as a nutrient transfer hub to nematode</div>
💀 EFFECT ON PLANT:
<div style="margin-bottom: 6px; padding-left: 20px; text-indent: -20px;"><strong style="color:#d35400; margin-right: 6px;">—</strong>Vascular tissue disrupted: water & nutrient transport blocked</div><div style="margin-bottom: 6px; padding-left: 20px; text-indent: -20px;"><strong style="color:#d35400; margin-right: 6px;">—</strong>5–7 giant cells per nematode head</div><div style="margin-bottom: 6px; padding-left: 20px; text-indent: -20px;"><strong style="color:#d35400; margin-right: 6px;">—</strong>Gall tissue prevents normal root function</div><div style="padding-left: 20px; text-indent: -20px;"><strong style="color:#d35400; margin-right: 6px;">—</strong>Secondary pathogens exploit weakened tissue</div>
Giant cells are unique to root-knot nematode infection — not found in any other nematode species
Economic Impact of Nematodes on Crops
ECONOMIC IMPACT ON GLOBAL AGRICULTURE
$80–173 Billion
Annual global crop losses due to nematodes
21% Average Yield Loss
Across major crop species worldwide
4,100+ Species
Plant-parasitic nematode species identified
CROP-SPECIFIC YIELD LOSSES
Up to 80% yield loss
41–88% storage root loss
10–30% reduction
10–25% loss
Significant losses from Radopholus similis
DIRECT LOSSES
Reduced yield, lower crop quality, and complete loss of marketability.
INDIRECT LOSSES
Increased pesticide reliance, exorbitant soil remediation costs, and severe disease synergy with pathogenic fungi and bacteria.
Climate change is expanding nematode-favorable conditions globally
MANAGEMENT & CONTROL STRATEGIES
Integrated Nematode Management (INM)
BIOLOGICAL CONTROL
Trichoderma spp., Purpureocillium lilacinus, Pasteuria penetrans. Nematophagous fungi and bacteria that parasitize nematode eggs and juveniles. Eco-friendly, sustainable option.
CHEMICAL NEMATICIDES
Carbofuran, Oxamyl, Fenamiphos. Applied to soil pre-planting. Highly effective but environmental concerns; restricted in many countries. Use with caution.
CROP ROTATION
Rotate with non-host crops (cereals, maize). Breaks nematode reproduction cycle. Reduces soil population by 60–80% over 2–3 seasons.
SOIL SOLARIZATION
Cover moist soil with clear polyethylene film for 4–6 weeks in summer. Solar heating kills nematodes and eggs in top 30 cm of soil.
RESISTANT VARIETIES
Use nematode-resistant crop cultivars (Mi-1 gene in tomato). Most sustainable long-term solution. Being enhanced through modern biotechnology.
Integrated Nematode Management combines multiple strategies for most effective, sustainable control
- nematodes
- plant-pathology
- meloidogyne-incognita
- nematology
- crop-science
- agriculture
- root-knot-nematode