Understanding Intramolecular Forces: Bonds & Examples
Explore the 4 main types of intramolecular forces: ionic, covalent, metallic, and coordinate bonding. Learn their roles in molecular structure and chemistry.
Intramolecular Forces
Understanding the Bonds That Hold Atoms Together
What are Intramolecular Forces?
Forces that act WITHIN a molecule.
They hold atoms together to form molecules or crystal lattices.
Determines the chemical identity of a substance.
Why Are They Important?
Intramolecular forces define how matter behaves. They control:
Molecular Structure & Geometry
Chemical Stability
Reactivity (How substances react)
The 4 Main Types
Ionic Bonding
Covalent Bonding
Metallic Bonding
Coordinate Bonding
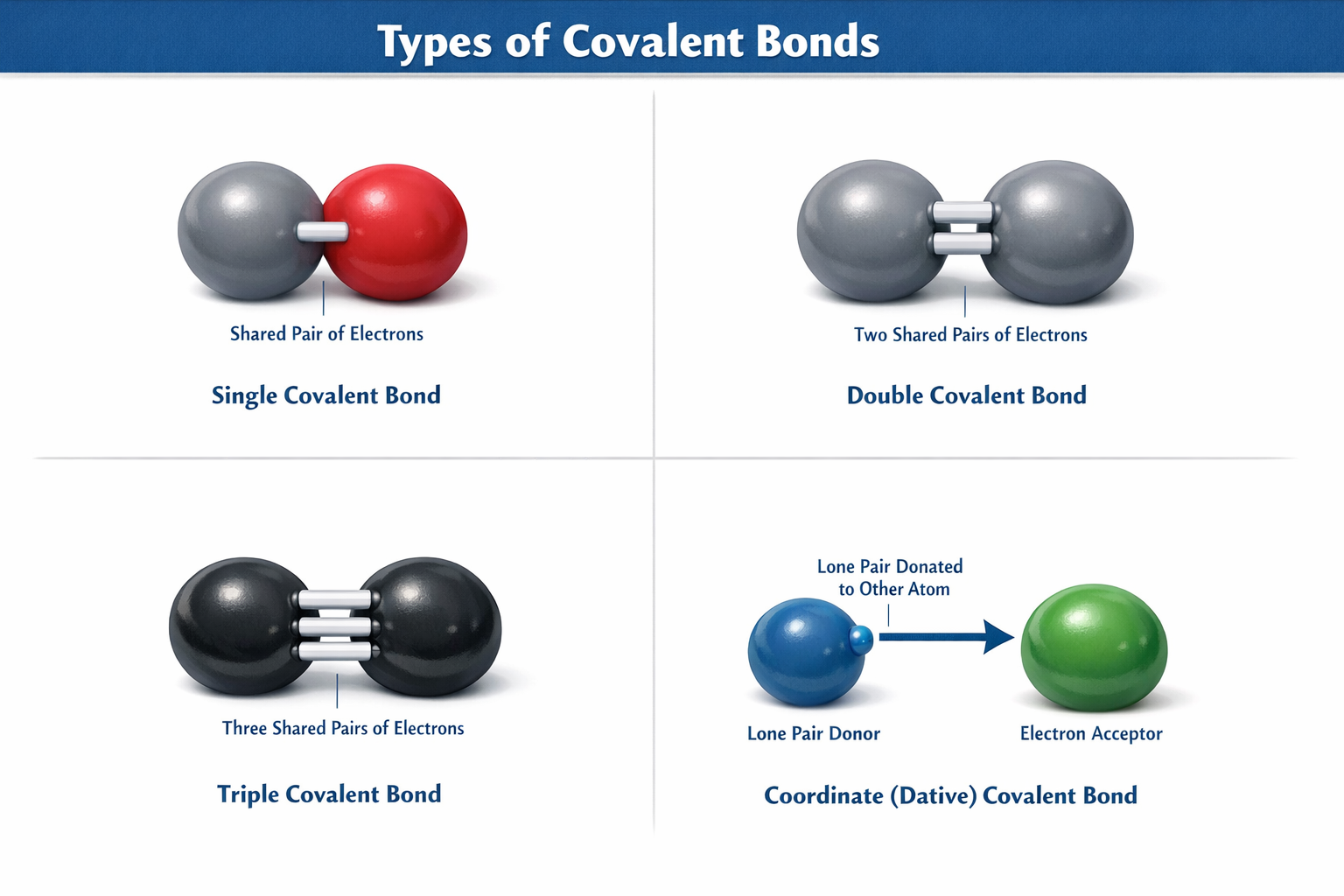
1. Covalent Bonding
Atoms SHARE pairs of electrons to become stable.
Types of Covalent Bonds
Nonpolar
Equal sharing of electrons. Example: H-H, O=O
Polar
Unequal sharing. One atom pulls harder. Example: H2O
Bond Strength Comparison
Triple bonds share 3 pairs of electrons, making them the strongest and shortest.
2. Ionic Bonding
One atom COMPLETELY TRANSFERS electrons to another.
Creates oppositely charged ions (+ and -)
Held together by electrostatic attraction
3. Metallic Bonding
Attraction between positive metal ions and a 'sea' of free-floating electrons.
Conducts electricity
Malleable and Ductile
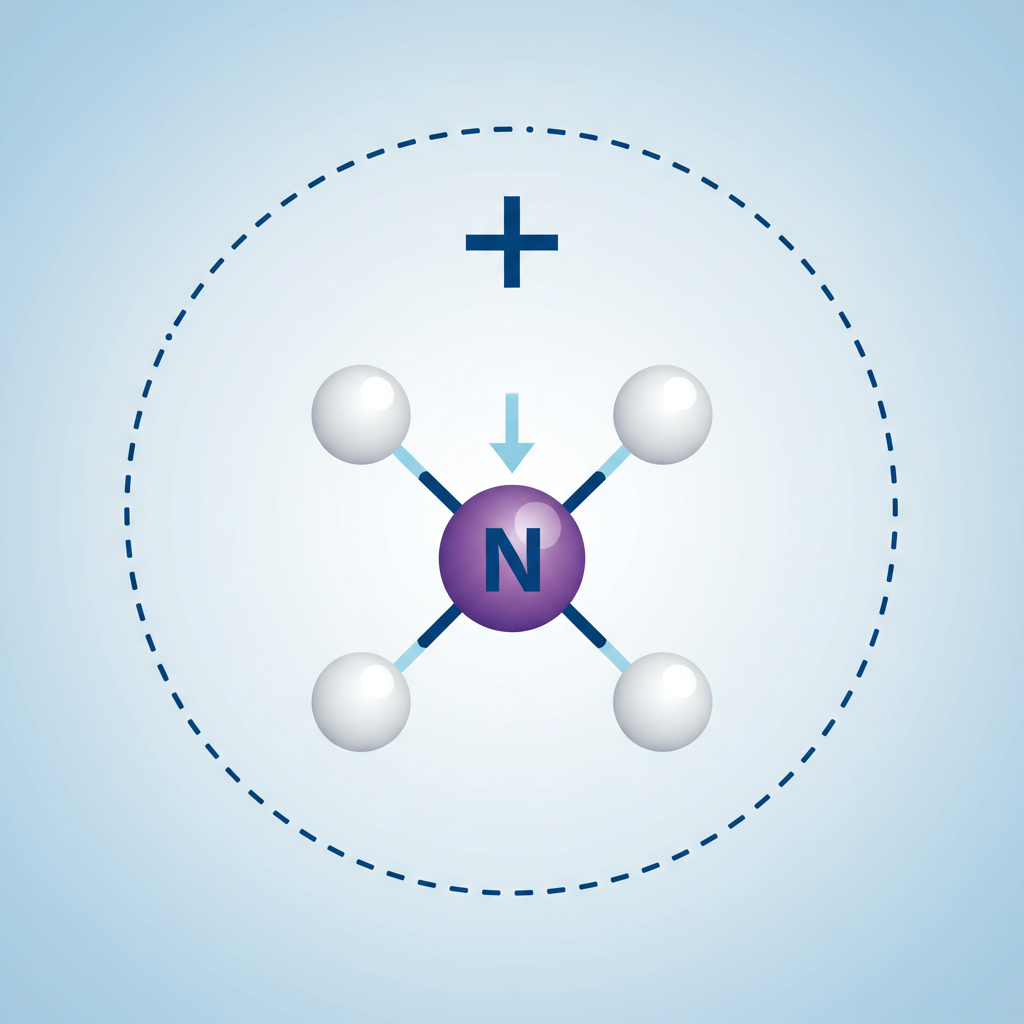
4. Coordinate Covalent Bond
A special covalent bond where BOTH shared electrons come from the SAME atom.
Example: Ammonium Ion (NH4+)
Intramolecular vs. Intermolecular
INTRAmolecular
Within the molecule. Very Strong. Example: Covalent Bond.
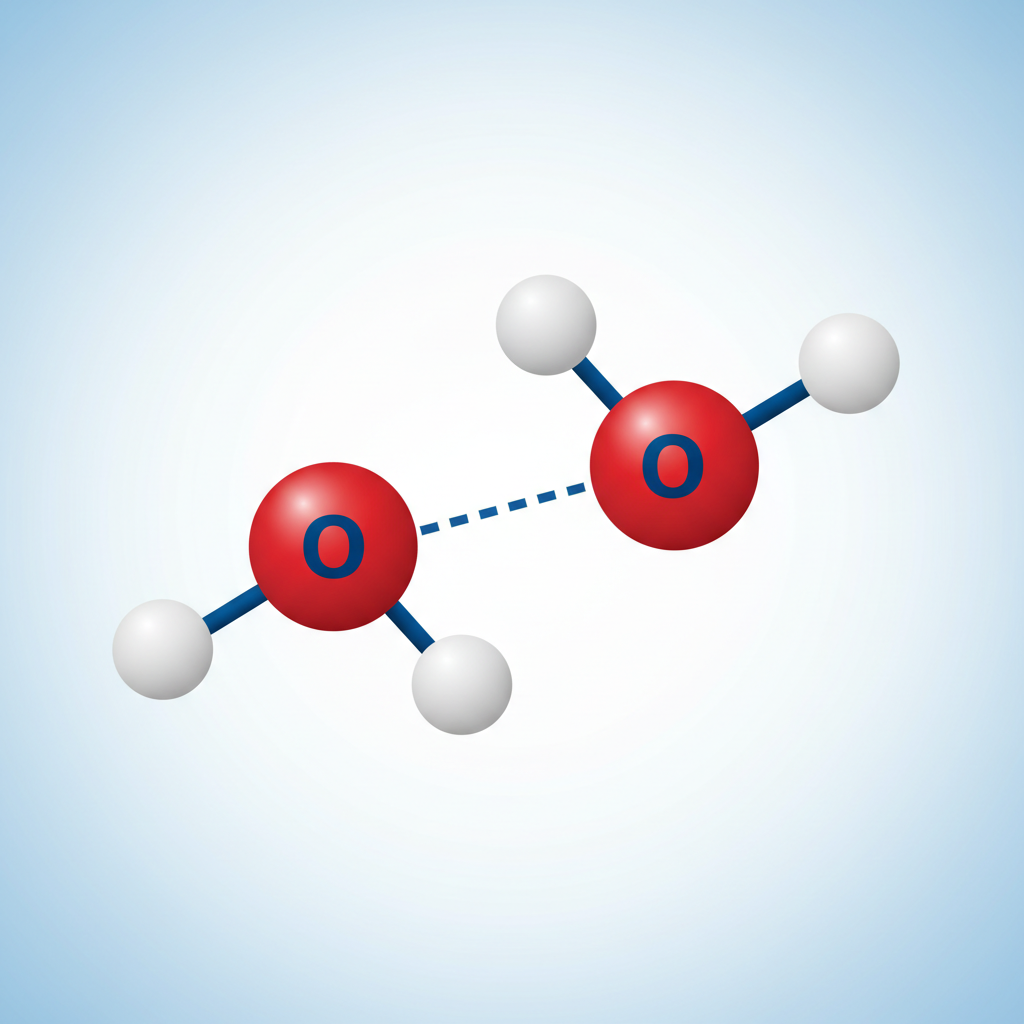
INTERmolecular
Between molecules. Weak. Example: Hydrogen Bond.
Real World Examples
Water (Covalent)
Table Salt (Ionic)
Copper Wire (Metallic)
Conclusions
Fundamental forces holding atoms together.
Determine chemical identity and structure.
Much stronger than intermolecular forces.
Essential for Organic Chemistry and Material Science.
References
1. Zumdahl, S. S., & Zumdahl, S. A. (2010). Chemistry. 2. Pauling, L. (1960). The Nature of the Chemical Bond. 3. Khan Academy - Intramolecular Forces Courses. 4. ScienceDirect - Chemical Bonding Articles.
Thank You!
Any Questions?
- chemistry
- intramolecular-forces
- covalent-bonding
- ionic-bonding
- metallic-bonding
- science-education
- molecular-structure
- chemical-bonds