PFR Design for Toluene Synthesis: Calculations & Balance
A technical guide on Toluene synthesis in a PFR, covering stoichiometry, molar mass, material balance, and reactor volume design equations.
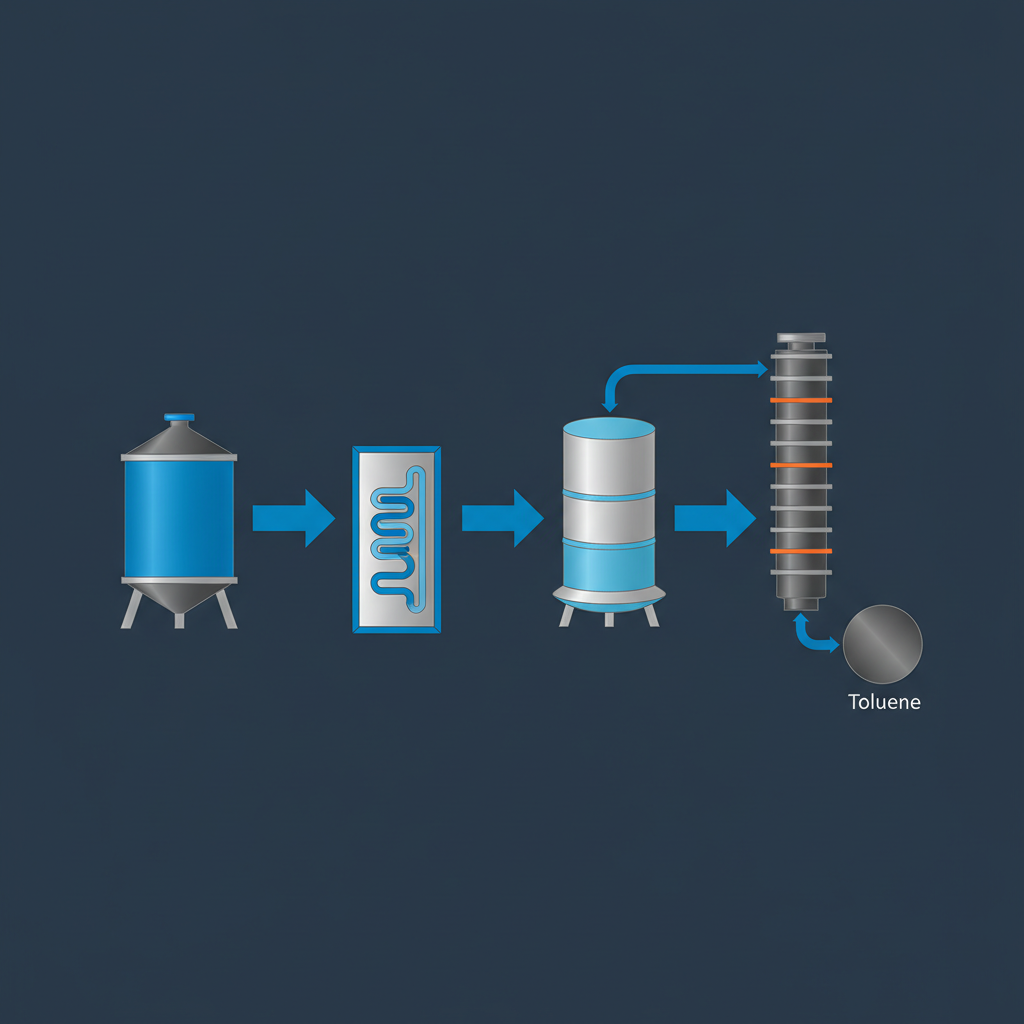
Toluene Synthesis in PFR
Design Calculation & Separation Process
Main Reaction Stoichiometry
C₆H₆ + CH₃OH → C₇H₈ + H₂O
Molar Masses (kg/kmol)
Target: 1000 kg Toluene<br><br>n(Tol) = m / M<br>n(Tol) = 1000 / 92<br>n(Tol) = 10.87 kmol
Assume Conversion (X) = 0.80
Required Reactant Benzene (nA0)
nA0 = n(Tol) / X
nA0 = 10.87 / 0.80 = 13.59 kmol
Input Mass Flow (kg)
Material Balance Output
Toluene (Product): 1000 kg
Water (By-product): 196 kg
Unreacted Benzene: 212 kg
dFA / dV = rA
PFR Design Equation
Integrated Volume Equation
V = ( FA0 / (k · CA0) ) · ln( 1 / (1 − X) )
Parameters
FA0 = 3.77 × 10⁻³ kmol/s
k = 5.56 × 10⁻⁵ s⁻¹
CA0 = 2.0 kmol/m³
Solution:<br><br>V = 55 m³
Thermal Balance (Exothermic)
Residence Time Calculation
Q (Volumetric Flow) = 1.89 × 10⁻³ m³/s
τ = V / Q
τ ≈ 2.9 × 10⁴ s ≈ 8.0 hours
Conclusion
Reactor Volume: 55 m³ Residence Time: 8 Hours Product: 1000 kg Toluene
- chemical-engineering
- pfr-design
- toluene-synthesis
- material-balance
- reactor-sizing
- stoichiometry