Phase Contrast Microscopy: Visualizing Living Cells
Learn the principles, history, and applications of phase contrast microscopy. Discover how Frits Zernike's Nobel Prize-winning technique visualizes live cells.
Phase Contrast Microscopy
Visualizing Transparent Specimens Without Staining
The Visibility Challenge
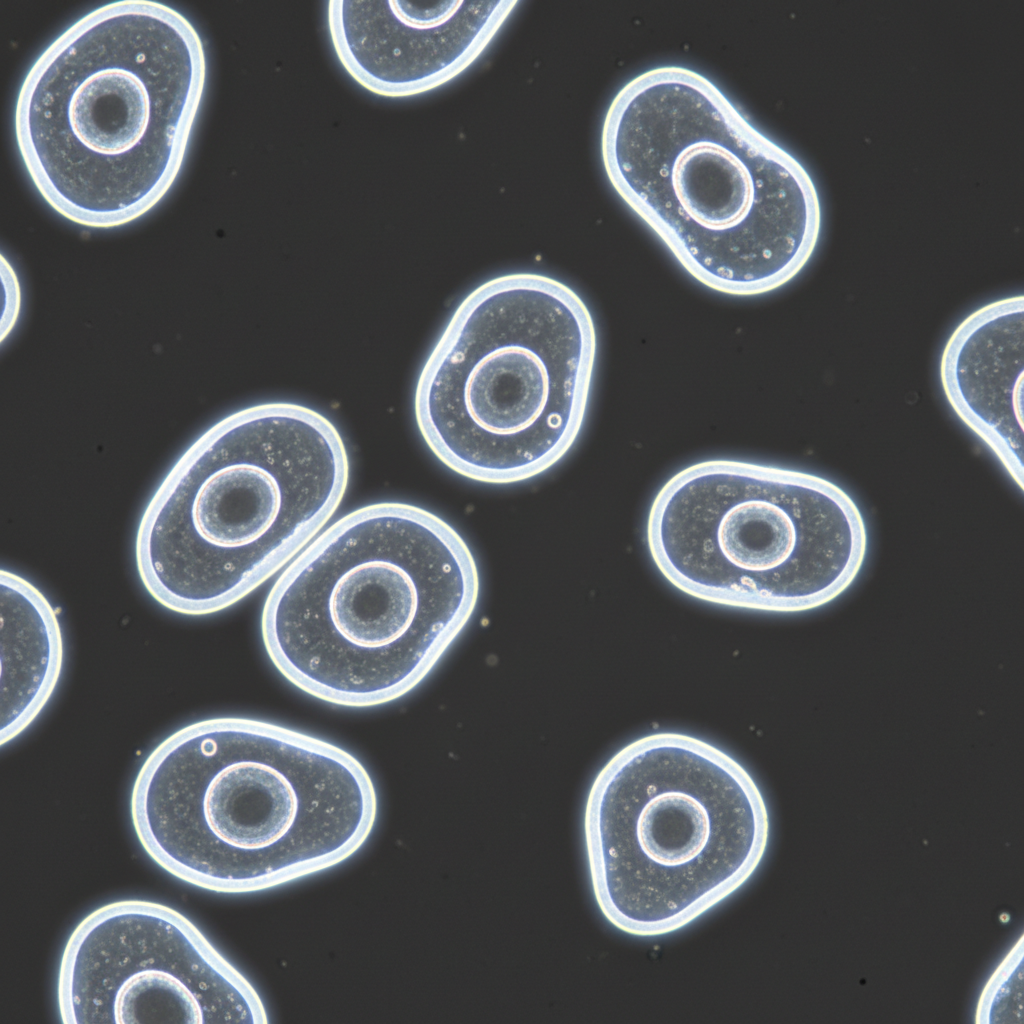
In standard bright-field microscopy, transparent specimens like living cells are often invisible. Because they do not absorb much light, there is little amplitude contrast between the specimen and the surrounding medium. Historically, this required killing and staining cells, which introduced artifacts and prevented the study of live dynamics.
History & Invention
The phased contrast technique was developed by Dutch physicist Frits Zernike in the 1930s. His discovery that phase changes in light could be converted into amplitude changes revolutionized biology. Zernike was awarded the Nobel Prize in Physics in 1953 for this breakthrough, enabling the detailed study of internal cell structures without chemical fixation.
Core Principles
Phase Objects: Biological structures change the phase of light passing through them due to refractive index differences.
Invisible to Visible: The human eye detects intensity (amplitude), not phase shifts.
Interference: The microscope converts phase shifts (~1/4 wavelength) into brightness variations via interference.
Result: High-contrast images where dense structures appear darker against a gray background.
Refractive Index (n) of Biological Materials
Phase contrast relies on the difference in refractive index between cell components and the medium. Even slight differences (e.g., Nucleus vs Cytoplasm) create the phase shift necessary for contrast.
Key Optical Components
1. Annular Diaphragm: Located in the condenser, it produces a hollow cone of light. 2. Phase Plate: Located in the objective lens, it has a ring that advances or retards the direct light by 1/4 wavelength. Alignment of these two rings is critical for the technique to function.
Visual Appearance: The Halo Effect
Mechanism: Light Inteference
Separation: Light is split into Direct Light (Surround) and Diffracted Light (Particle).
Phase Shift: Diffracted light is retarded by the specimen (~1/4 λ). The Phase plate shifts Direct light by another 1/4 λ.
Recombination: When waves recombine at the image plane, they are now 1/2 wavelength out of phase.
Destructive Interference: This causes destructive interference, making the specimen appear dark against a light background.
Phase contrast is the standard method for observing living cells in tissue culture. It transforms small phase differences into visible intensity differences.
Key Applications: Cell Culture, Sperm analysis, Silica dust monitoring
Advantages & Limitations
Advantages
Limitations
• Observe living cells in natural state • No staining or fixation required • High contrast for internal organelles • Combine with fluorescence
• Halo artifacts around thick specimens • Reduced resolution compared to DIC • Not suitable for thick samples • More expensive objectives
- microscopy
- biology
- cell-culture
- scientific-imaging
- frits-zernike
- lab-techniques
- biomedical-science