Intergenerational Trauma & Epigenetics: Molecular Memory
Explore how ancestral trauma affects gene expression through DNA methylation and histone modification across generations based on recent research.
Intergenerational Effects of Ancestral Trauma
Epigenetic Mechanisms and Human Manifestations
Based on the Review Article by Kaoutar Ousmoi (2026)
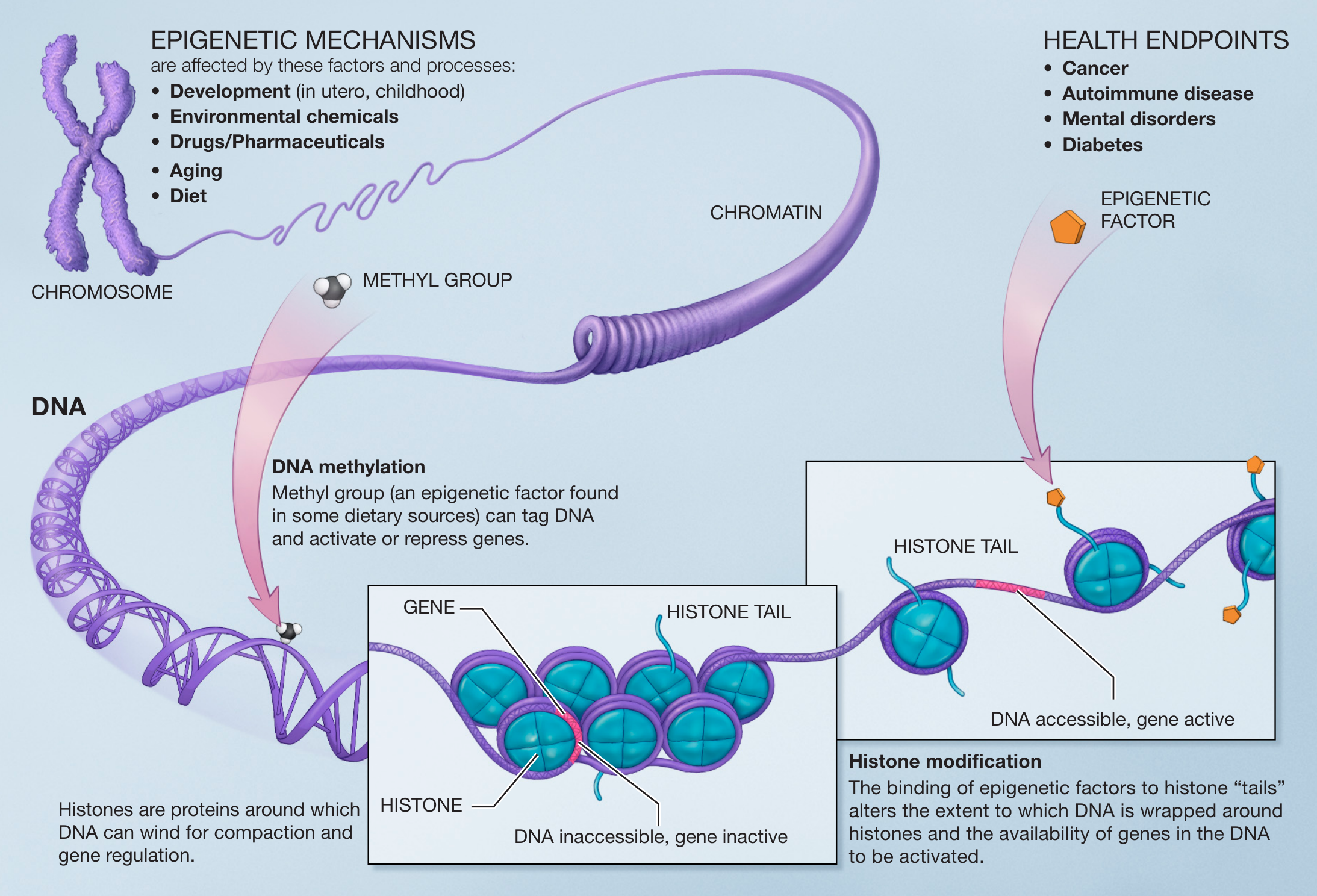
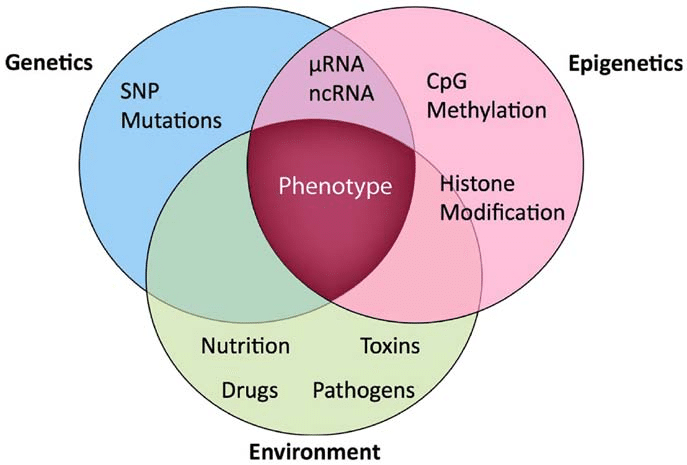
What is Epigenetics?
Study of how gene activity is altered and maintained without changing the primary DNA sequence.
Involves chemical modifications to DNA, chromatin structure, and RNA-based regulators.
Determines which genes are expressed ('turned on' or 'off'), when, and at what level.
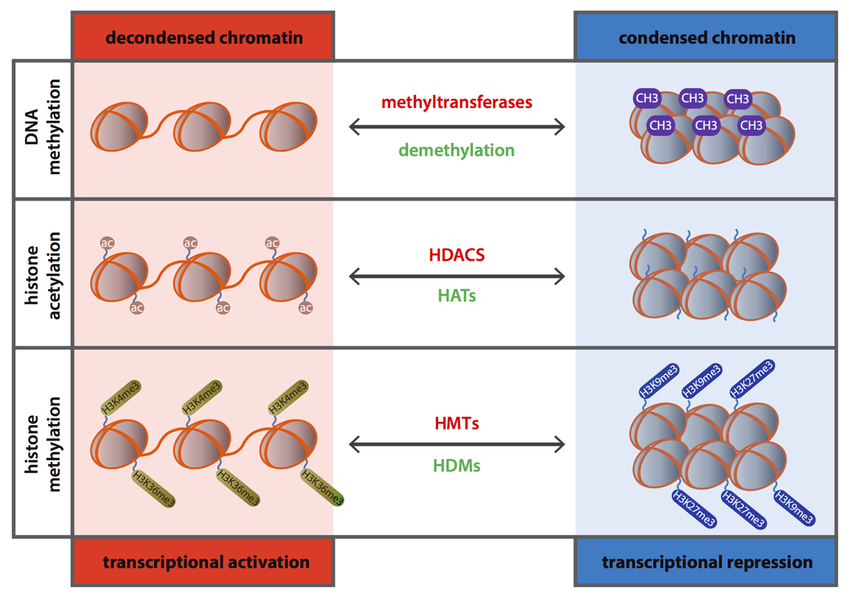
The Molecular Mechanisms
DNA Methylation
Addition of a methyl group (CH3) to Cytosine. Acts like a 'cap' that usually silences gene expression by making transcription difficult.
Histone Modification
Chemical changes to 'tails' of histone proteins. Affects how tightly DNA is wrapped; tighter wrapping = less accessible genes.
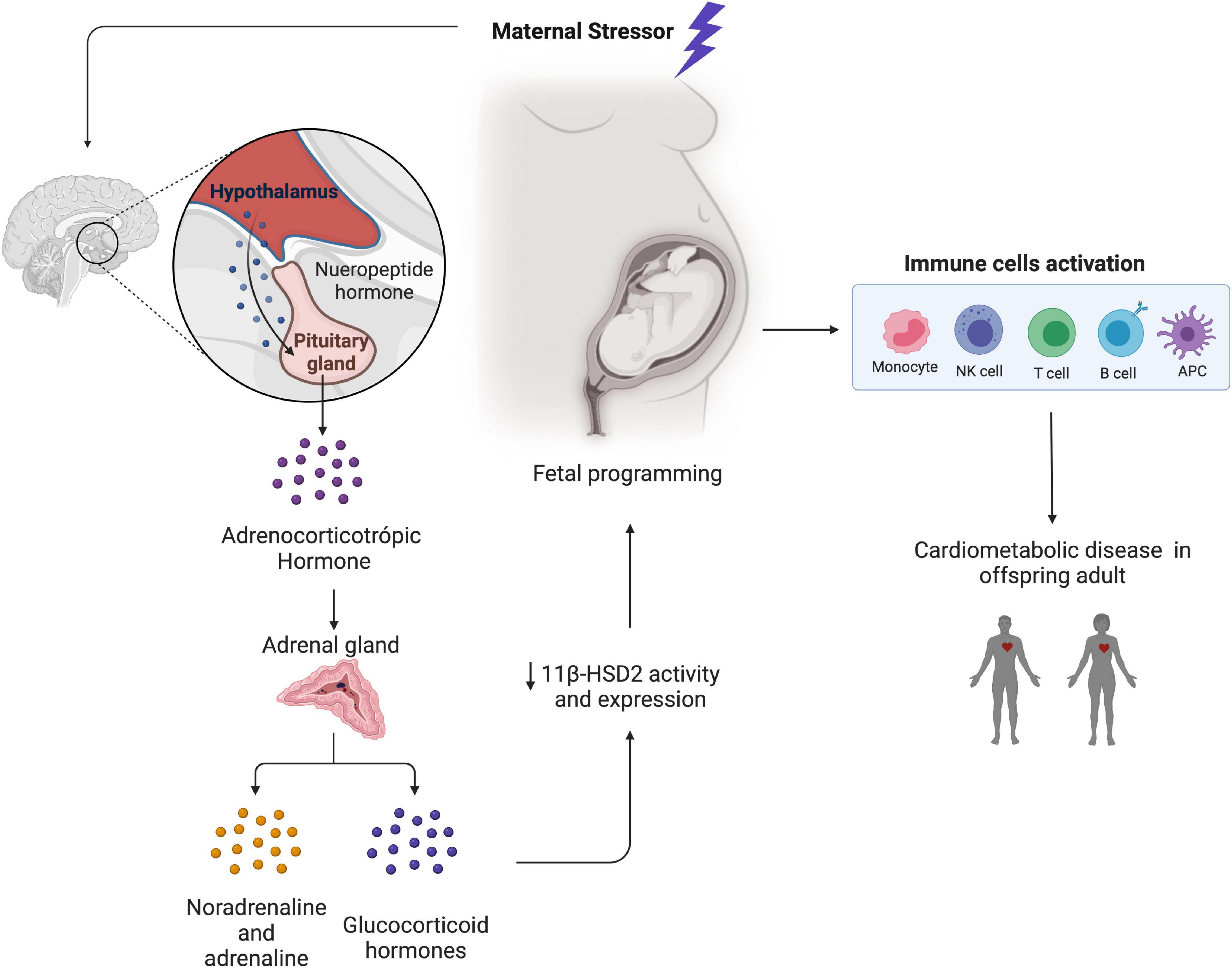
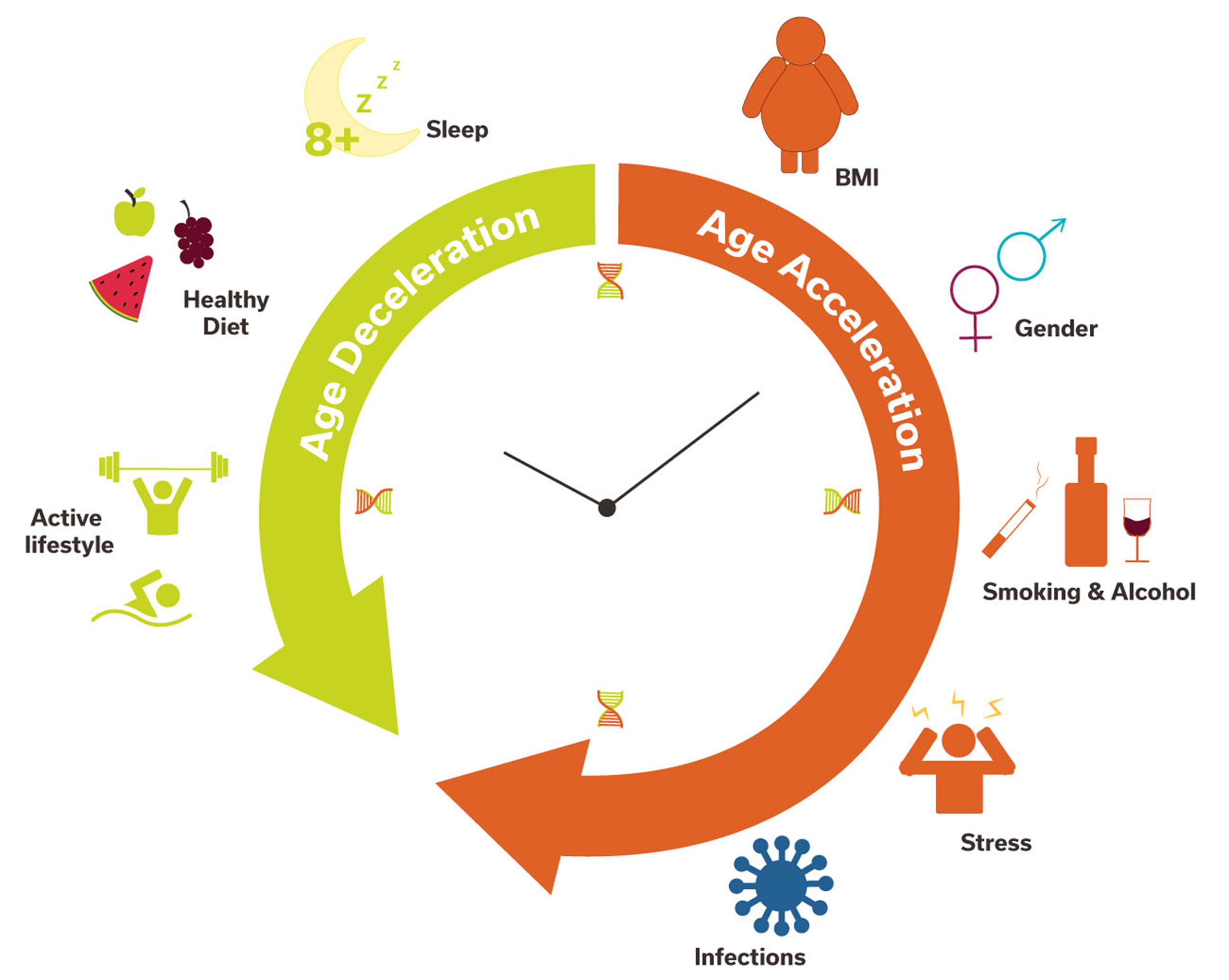
From Environment to Genome
External Stress / Trauma
Release of Cortisol & Cytokines
Intracellular Signaling
Epigenetic Enzymes (Writers & Erasers) Modify DNA/Histones
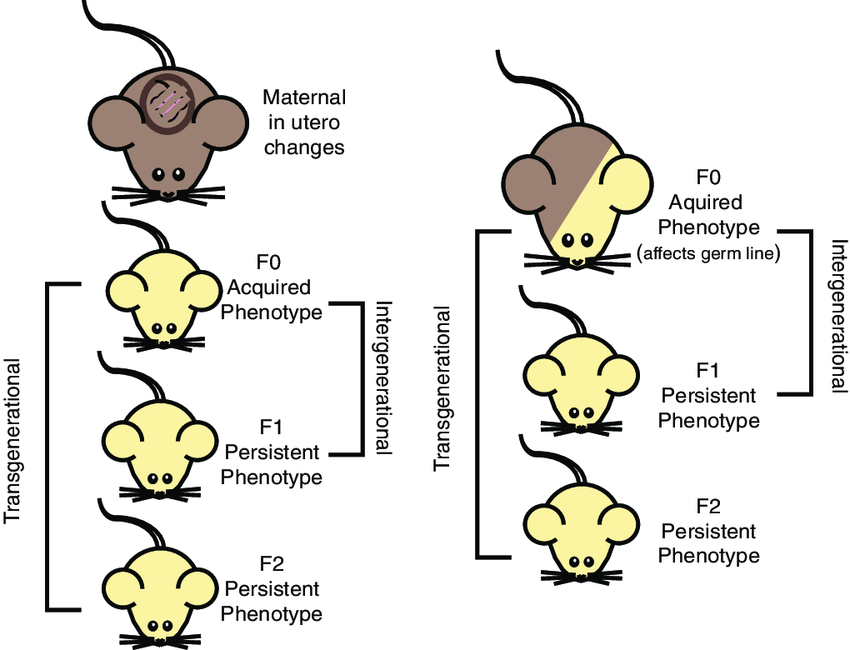
Transmission Route 1: Developmental Programming
Maternal trauma during pregnancy disrupts the HPA (stress-response) axis. Hormones cross the placenta, modifying fetal epigenetic marks.
Example: Increased methylation of the NR3C1 gene in children of mothers with depression, leading to altered stress reactivity.
Transmission Route 2: Germline Mediated
Can trauma affect sperm or egg cells before conception?
Animal studies show strong evidence. Mice conditioned to fear an odor showed reduced DNA methylation at specific receptor genes in sperm.
Result: Offspring inherited sensitivity to that specific odor without ever experiencing it themselves.
Human Evidence: Individual Manifestations
Holocaust Survivor Study: FKBP5 Gene Methylation
Study examined the FKBP5 gene (regulates stress response/cortisol).
Specific methylation site (intron 7) showed inverse correlation between survivors and offspring.
Community Level: The Shadow of Genocide
Studies on descendants of the 1994 Genocide against the Tutsi in Rwanda.
Mothers exposed to severe violence during pregnancy had children with altered methylation in brain/stress genes.
Accelerated Epigenetic Aging: Descendants showed 'older' biological age than chronological age.
Multigenerational Impact: Syrian Refugee Families
Research identified 35 CpG sites associated with war violence. In 32 of these, methylation shifted in the SAME direction across 3 generations (Grandparent -> Parent -> Child), regardless of when exposure occurred.
The Biosocial Loop: Reinforcement
Biology does not exist in a vacuum. Inherited vulnerabilities are often reinforced by the post-natal environment.
Inherited Epigenetic Marks (Biological Vulnerability) -> High-Stress Social Environment (Hypervigilant Caregivers) -> Recurrent Activation of Stress System -> Further Reinforcement of Methylation Patterns
Discussion & Critical nuance
Correlation vs. Causation
Human studies are largely correlational. It is difficult to distinguish biological transmission from the effects of shared environments and parenting practices.
Risk of Over-Biologizing
Focusing solely on 'damaged biology' can distract from structural causes (poverty, racism, war). Interpretations must remain biosocial.
Conclusion
Extreme adversity leaves molecular traces in stress systems (epigenetics) that can persist across generations.
Epigenetic inheritance is not 'destiny' or fixed damage. It is a modifiable component of a broader process involving social conditions, policy, and care.
- epigenetics
- intergenerational-trauma
- dna-methylation
- genetics
- psychology
- biology
- molecular-biology