Industrial Aluminum Production: Bayer & Hall-Héroult Processes
Explore the complete journey of aluminum from bauxite ore to metal, covering chemical foundations, the electrolysis mechanism, and energy recycling benefits.
Industrial Aluminum Production
From Bauxite to Metal: The Bayer & Hall-Héroult Processes
The Raw Material: Bauxite
Primary source: Bauxite ore containing Aluminum Oxide (Al2O3).
Impurities: Iron oxides (red mud) and silicates must be removed.
Weathering process: Formed in tropical regions via silicate weathering.
Chemical Foundations: Amphoteric Behavior
Aluminum Hydroxide Al(OH)3 exhibits amphoteric properties, dissolving in both acids and strong bases. This property is crucial for the Bayer Process to separate pure Alumina from impurities.
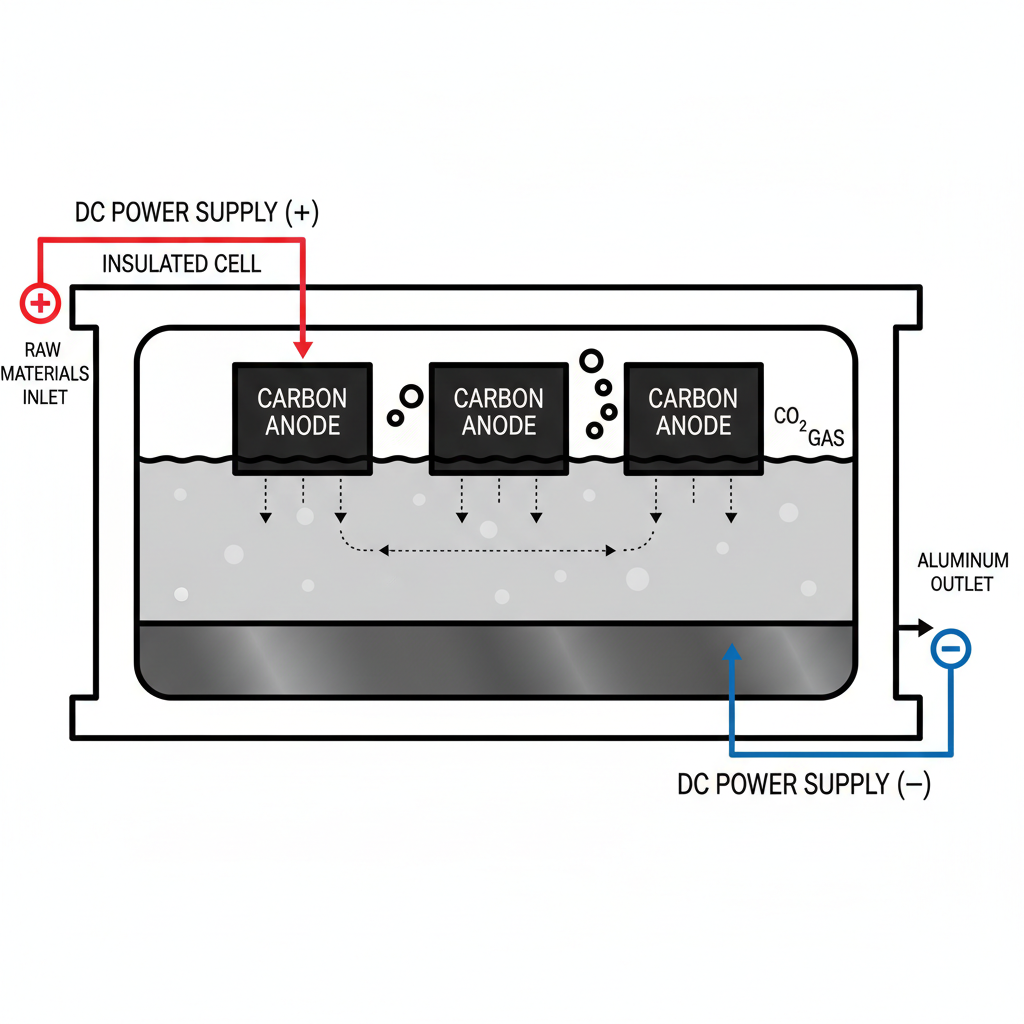
The Hall-Héroult Electrolysis Process
Produces 99.5-99.8% pure aluminum. Alumina (Al2O3) is dissolved in molten Cryolite (Na3AlF6) at approx. 960°C to lower the melting point.
Electrolysis Mechanism
Cathode: Liquid Aluminum forms at the bottom of the cell. Reaction: Al³⁺ + 3e⁻ → Al(l)
Anode: Carbon blocks react with oxygen. Reaction: 2O²⁻ + C(s) → CO2(g) + 4e⁻
Note: Anodes are consumed during the process and must be regularly replaced along with Fluoride salts.
Energy Consumption Analysis
Comparison of energy required for primary production versus recycling. Recycling requires only ~5% of the original energy input.
Environmental Challenges
CO₂ Emissions
Anode consumption generates significant CO₂. Primary production has a high carbon footprint.
PFC Emissions
Occasional "anode effects" release CF4 and C2F6, potent greenhouse gases.
Red Mud
Iron-rich waste from the Bayer process poses storage and alkalinity hazards.
Fluoride Limits
Strict controls required to prevent hydrogen fluoride release into local ecosystems.
Important Compounds: Aluminum Chloride
Aluminum halides, specifically AlCl3, play a critical role in organic chemistry. AlCl3 acts as a strong Lewis Acid catalyst.
Application: Friedel-Crafts alkylation and acylation. Structure: Exists as a dimer (Al2Cl6) in gas phase or non-polar solvents.
Strategic Outlook
Recycling is the Future: Due to the high energy cost of primary production (approx 15 kWh/kg), recycling provides huge economic margin.
Material Properties: Lightweight, corrosion-resistant (due to protective oxide layer), and high conductivity ensure growing demand in EV and aerospace sectors.
Summary
From ore to innovation
- aluminum-production
- bayer-process
- hall-heroult
- electrolysis
- bauxite
- industrial-chemistry
- metal-recycling