Managing Refractory Diarrhea in HER2+ Breast Cancer Patients
Explore a clinical case study on managing refractory diarrhea and toxicity caused by Pertuzumab and Docetaxel in breast cancer treatment.
Management of Refractory Diarrhoea in Breast CA
Case Discussion
Clinical Standards: UKONS, CCO, and BC Cancer Toxicity Management Guidelines
Baseline & GI Vulnerability
<ul><li><strong>Diagnosis:</strong> HER2+ Breast Cancer (ER 0, PR 4)</li><li><strong>Regimen:</strong> TCHP-P (Docetaxel, Carboplatin, Pertuzumab, Trastuzumab)</li><li><strong>History:</strong> Chronic Grade 2 diarrhea (5–6 stools/day) requiring dose reductions every cycle.</li></ul>
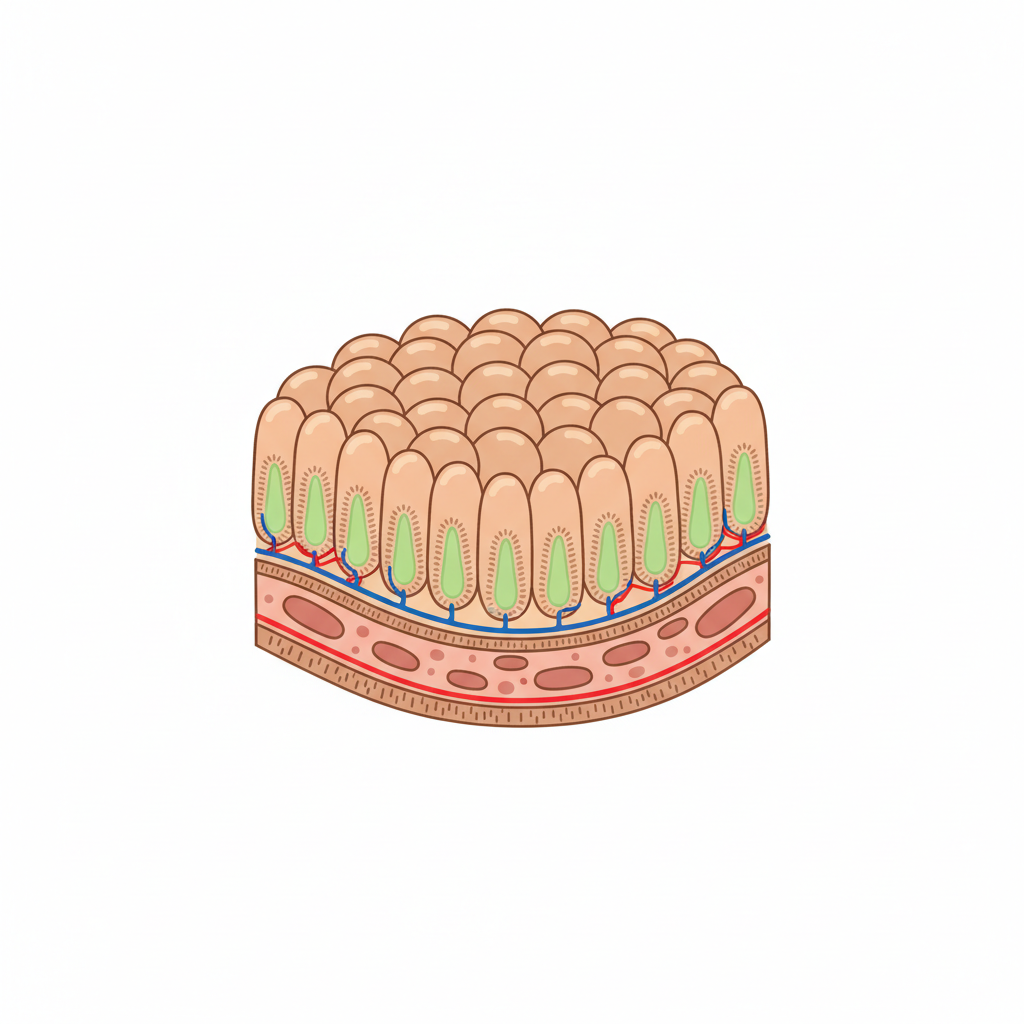
<strong>The 'Pertuzumab Effect':</strong> Inhibits HER2/EGFR on intestinal enterocytes. This impairs mucosal repair mechanisms, thinning the gut lining and priming it for fulminant infection.
Teaching Point: The 'Double-Hit' Toxicity
Phesgo (Pertuzumab)
Docetaxel (Taxane)
<ul><li><strong>Mechanism:</strong> HER2 inhibition disrupts electrolyte transport & mucosal repair.</li><li><strong>Effect:</strong> Secretory diarrhea (High Volume).</li><li><strong>Onset:</strong> Early (Hours/Days).</li><li><strong>Course:</strong> Prolonged/Chronic.</li></ul>
<ul><li><strong>Mechanism:</strong> Antimitotic toxicity to rapidly dividing crypt cells.</li><li><strong>Effect:</strong> Mucositis & Epithelial sloughing.</li><li><strong>Onset:</strong> Nadir (Day 7–14).</li><li><strong>Course:</strong> Transient (7–10 days).</li></ul>
Result: Watery secretory output + Sloughing of protective barrier = High risk of severe colitis & CDI.
Acute Presentation: Septic Shock (Day 0)
BP 80/47 mmHg | HR: Tachycardic | Temp: 38.5°C
<ul><li><strong>AKI Stage 3:</strong> eGFR 12 (Baseline 84)</li><li><strong>WBC:</strong> 16.7</li><li><strong>CRP:</strong> 403 mg/L</li></ul>
Immediate Resuscitation + IV Ceftazidime (Patient Penicillin Intolerant).
Timeline: Day +10 Post-Chemotherapy Nadir
Clinical Course: The 'Flare'
<ul><li><strong>Day +3:</strong> Transient improvement. AKI resolved (eGFR 84).</li><li><strong>Day +4 (The Flare):</strong> Sudden WBC spike to 30.8. AXR performed to rule out toxic megacolon (Negative for free air, confirmed thickening).</li><li><strong>Day +5:</strong> CRP peaked at 295. Escalation to IV Metronidazole and Fidaxomicin.</li></ul>
Why is the Patient Still Sick? (The Paradox)
<strong>The "Paradox" Explained:</strong><br><br><span style="color:#28A745; font-weight:bold;">✔ The Blood is Better:</span><br>The antibiotics successfully killed the bacteria. The infection is gone.<br><br><span style="color:#FF4D4D; font-weight:bold;">✘ The Gut Needs Time:</span><br>While the bacteria are dead, the gut lining is still healing from the inflammation. It temporarily loses its ability to absorb liquid, causing ongoing symptoms despite the cure.<br><br><strong>Key Takeaway:</strong> We fixed the infection, but the tissue recovery lags behind.
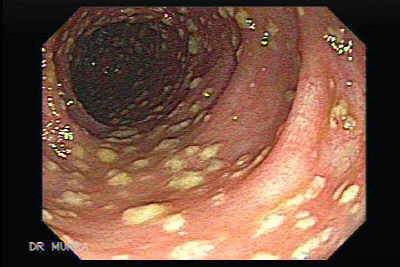
Workup: Imaging & Endoscopy
<strong>CT CAP:</strong> Confirmed widespread Diffuse Colitis; ruled out perforation.
<strong>Flexi-Sigmoidoscopy:</strong> Visual confirmation of Pseudomembranous Colitis.
<strong>Note on Diagnostics:</strong> Per BC Cancer/CCO guidelines, visual confirmation of pseudomembranes supersedes negative toxin assays. High transit volume from Phesgo likely causes toxin dilution, leading to false negatives.
Differential Diagnosis: Ruling Out CMV
<ul><li><strong>Status:</strong> Weakly positive CMV IgM.</li><li><strong>Pending:</strong> CMV DNA PCR (Viral Load).</li></ul>
Given the lack of clinical improvement despite antibiotic therapy and normal CRP, CMV reactivation must be ruled out via PCR before starting anti-secretory agents or steroids.
Management: Antibiotic Strategy Evolution
<strong>Phase 1: Initial Escalation</strong><br>12 days Fidaxomicin + 13 days IV Metronidazole.<br><em>Outcome:</em> Cleared systemic inflammation (Normal CRP), but mucosal disease persisted.
<strong>Phase 2: High-Dose Vancomycin</strong><br>Started Day +17 (250 mg Q4H).
<strong>Phase 3 (Current): Extended Fidaxomicin</strong><br>200 mg BD (5 days) → 200 mg OD alternate days (until Day 21).<br><em>Rationale:</em> Protect gut while mucosa regenerates. Patient not a candidate for FMT.
Supportive Care: Electrolyte Management
<strong>The Challenge:</strong> Persistent high-volume GI losses (18–22 episodes/day) leading to refractory low Magnesium and Potassium levels.
<strong>Management:</strong><br>- Daily IV replacement required to maintain cardiac stability.<br>- Renal function remains stable (eGFR 84).
Management Algorithm (BC Cancer/UKONS)
1. <strong>CDI Control:</strong> Complete the 21-day Fidaxomicin pulse/taper.
2. <strong>CMV Check:</strong> Await PCR. If positive → Initiate Ganciclovir.
3. <strong>Anti-Secretory:</strong> If CMV is negative and output remains refractory, consult Gastro regarding Octreotide.
4. <strong>Supportive:</strong> Twice daily electrolyte checks and aggressive IV replacement.
Synthesis & Conclusion
<strong>Achievement:</strong> Successful stabilization of Septic Shock and full recovery of AKI Stage 3 (eGFR 12 to 84).
<strong>Current Stagnation:</strong> Systemic markers are normal, but local mucosal failure persists due to the 'Double-Hit' of Phesgo and Docetaxel toxicity.
<strong>Next Steps:</strong><br>1. Chase CMV PCR result.<br>2. Continue daily blood monitoring.<br>3. Extended antibiotic taper to allow mucosal regeneration.
Questions?
References
<ul><li>UKONS (UK Oncology Nursing Society) Acute Oncology Guidelines.</li><li>CCO (Cancer Care Ontario) Symptom Management Guidelines.</li><li>BC Cancer Toxicity Management Standards.</li></ul>
- breast-cancer
- oncology
- clinical-case
- toxicity-management
- chemotherapy-side-effects
- pertuzumab
- medical-education